08/01/2019
TXA à l'avant: Peut être pas si simple !
Prehospital Tranexamic Acid Administration During Aeromedical Transport After Injury.
Tranexamic acid (TXA) has been shown to reduce mortality in the treatment of traumatic hemorrhage. This effect seems most profound when given early after injury. We hypothesized that extending a protocol for TXA administration into the prehospital aeromedical setting would improve outcomes while maintaining a similar safety profile to TXA dosed in the emergency department (ED).
MATERIALS AND METHODS:
We identified all trauma patients who received TXA during prehospital aeromedical transport or in the ED at our urban level I trauma center over an 18-mo period. These patients had been selected prospectively for TXA administration using a protocol that selected adult trauma patients with high-risk mechanism and concern for severe hemorrhage to receive TXA. Patient demographics, vital signs, lab values including thromboelastography, blood administration, mortality, and complications were reviewed retrospectively and analyzed.
RESULTS:
One hundred sixteen patients were identified (62 prehospital versus 54 ED). Prehospital TXA patients were more likely to have sustained blunt injury (76% prehospital versus 46% ED, P = 0.002). There were no differences between groups in injury severity score or initial vital signs.
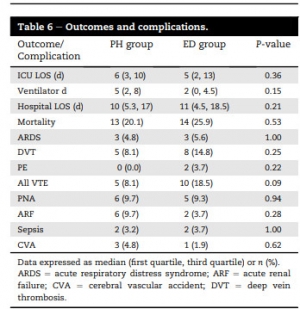
There were no differences in complication rates or mortality. Patients receiving TXA had higher rates of venous thromboembolic events (8.1% in prehospital and 18.5% in ED) than the overall trauma population (2.1%, P < 0.001).
CONCLUSIONS:
Prehospital administration of TXA during aeromedical transport did not improve survival compared with ED administration. Treatment with TXA was associated with increased risk of venous thromboembolic events. Prehospital TXA protocols should be refined to identify patients with severe hemorrhagic shock or traumatic brain injury.
30/12/2018
HEXACYL: Oui , mais attention à la MTE
Tranexamic acid administration is associated with an increased risk of posttraumatic venous thromboembolism.
BACKGROUND:
Tranexamic acid (TXA) is used as a hemostatic adjunct for hemorrhage control in the injured patient and reduces early preventable death. However, the risk of venous thromboembolism (VTE) has been incompletely explored. Previous studies investigating the effect of TXA on VTE vary in their findings. We performed a propensity matched analysis to investigate the association between TXA and VTE following trauma, hypothesizing that TXA is an independent risk factor for VTE.
METHODS:
This retrospective study queried trauma patients presenting to a single Level I trauma center from 2012 to 2016. Our primary outcome was composite pulmonary embolism or deep vein thrombosis. Mortality, transfusion, intensive care unit and hospital lengths of stay were secondary outcomes. Propensity matched mixed effects multivariate logistic regression was used to determine adjusted odds ratio (aOR) and 95% confidence intervals (95% CI) of TXA on outcomes of interest, adjusting for prespecified confounders. Competing risks regression assessed subdistribution hazard ratio of VTE after accounting for mortality.
RESULTS:
Of 21,931 patients, 189 pairs were well matched across propensity score variables (standardized differences <0.2). Median Injury Severity Score was 19 (interquartile range, 12-27) and 14 (interquartile range, 8-22) in TXA and non-TXA groups, respectively (p = 0.19). Tranexamic acid was associated with more than threefold increase in the odds of VTE (aOR, 3.3; 95% CI, 1.3-9.1; p = 0.02).
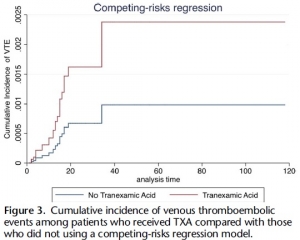
Tranexamic acid was not significantly associated with survival (aOR, 0.86; 95% CI, 0.23-3.25; p = 0.83). Risk of VTE remained elevated in the TXA cohort despite accounting for mortality (subdistribution hazard ratio, 2.42; 95% CI, 1.11-5.29; p = 0.03).
CONCLUSION:
Tranexamic acid may be an independent risk factor for VTE. Future investigation is needed to identify which patients benefit most from TXA, especially given the risks of this intervention to allow a more individualized treatment approach that maximizes benefits and mitigates potential harms.
| Tags : coagulopathie
15/12/2018
Hydroxocobalamine: Pour la coagulopathie et la pression
Intravenous Hydroxocobalamin Versus Hextend Versus Control for Class III Hemorrhage Resuscitation in a Prehospital Swine Model
Bebarta VS et Al. Mil Med. 2018 Nov 5;183(11-12):e721-e729.
Background: Hydroxyethyl starch (Hextend) has been used for hemorrhagic shock resuscitation, however, hydroxyethyl starch may be associated with adverse outcomes.
Objective: To compare systolic blood pressure (sBP) in animals that had 30% of their blood volume removed and treated with intravenous hydroxocobalamin, hydroxyethyl starch, or no fluid.
Methods: Twenty-eight swine (45-55 kg) were anesthetized and instrumented with continuous femoral and pulmonary artery pressure monitoring. Animals were hemorrhaged 20 mL/kg over 20 minutes and then administered 150 mg/kg IV hydroxocobalamin in 180 mL saline, 500 mL hydroxyethyl starch, or no fluid and monitored for 60 minutes. Data were modeled using repeated measures multivariate analysis of variance.
Results: There were no significant differences before treatment. At 20 minutes after hemorrhage, there was no significant difference in mean sBP between treated groups, however, control animals displayed significantly lower mean sBP (p < 0.001). Mean arterial pressure and heart rate improved in the treated groups but not in the control group (p < 0.02). Prothrombin time was longer and platelet counts were lower in the Hextend group (p < 0.05). Moreover, thromboelastography analysis showed longer clotting (K) times (p < 0.05) for the hydroxyethyl starch-treated group.
Conclusion: Hydroxocobalamin restored blood pressure more effectively than no treatment and as effectively as hydroxyethyl starch but did not adversely affect coagulation.
09/12/2018
Délivrance du sang: Merci les TAP
BACKGROUND:
Haemorrhagic shock remains the leading cause of preventable death in overseas and austere settings. Transfusion of blood components is critical in the management of this kind of injury. For French naval and ground military units, this supply often takes too long considering the short shelf-life of red blood cell concentrates (RBCs) and the limited duration of transport in cooling containers (five to six days). Air-drop supply could be an alternative to overcome these difficulties on the condition that air-drop does not cause damage to blood units.
METHODS:
After a period of study and technical development of packaging, four air-drops at medium and high altitudes were performed with an aircraft of the French Air Force. After this, one air-drop was carried out at medium altitude with 10 RBCs and 10 French lyophilised plasma (FLYP). A second air-drop was performed with a soldier carrying one FLYP unit at 12 000 feet. For these air-drops real blood products were used, and quality control testing and temperature monitoring were performed.
RESULTS:
The temperatures inside the containers were within the normal ranges. Visual inspection indicated that transfusion packaging and dumped products did not undergo deterioration. The quality control data on RBCs and FLYP, including haemostasis, suggested no difference before and after air-drop.
DISCUSSION:
The operational implementation of the air-drop of blood products seems to be one of the solutions for the supply of blood products in military austere settings or far forward on battlefield, allowing safe and early transfusion.
08/11/2018
Transport du sang en opex: Loin d'être anodin !
Bringing Packed Red Blood Cells to the Point of Combat Injury: Are We There Yet?
Ünlü et Al.Turk J Haematol. 2018 Aug 3;35(3):185-191.
-------------------------------
Transfuser le plus tôt possible serait un facteur de survie chez le blessé de guerre. Encore faut il disposer de sang et donc d'en transporter. On connaît les règles imposées en métropole. Les conditions de transport en opérations 'ont rien à voir avec celles de métropole. C'est bien ce que décrit cet article qui incite à la prudence. Seules 13% de poches récentes soumises à des vibrations intenses peuvent être transfusées après 06h d'exposition à des vibrations intenses.
-------------------------------
OBJECTIVE:
Hemorrhage is the leading cause of injury-related prehospital mortality. We investigated worst-case scenarios and possible requirements of the Turkish military. As we plan to use blood resources during casualty transport, the impact of transport-related mechanical stress on packed red blood cells (PRBCs) was analyzed.
MATERIALS AND METHODS:
The in vitro experiment was performed in the environmental test laboratories of ASELSAN®. Operational vibrations of potential casualty transport mediums such as Sikorsky helicopters, Kirpi® armored vehicles, and the NATO vibration standard MIL-STD-810G software program were recorded. The most powerful mechanical stress, which was created by the NATO standard, was applied to 15 units of fresh (≤7 days) and 10 units of old (>7 days) PRBCs in a blood cooler box. The vibrations were simulated with a TDS v895 Medium-Force Shaker Device. On-site blood samples were analyzed at 0, 6, and 24 h for biochemical and biomechanical analyses.
RESULTS:
The mean (±standard deviation) age of fresh and old PRBCs was 4.9±2.2 and 32.8±11.8 days, respectively. Six-hour mechanical damage of fresh PRBCs was demonstrated by increased erythrocyte fragmentation rates (p=0.015), hemolysis rates (p=0.003), and supernatant potassium levels (p=0.003) and decreased hematocrit levels (p=0.015). Old PRBC hemolysis rates (p=0.015), supernatant potassium levels (p=0.015), and supernatant hemoglobin (p=0.015) were increased and hematocrit levels were decreased (p=0.015) within 6 h.

Two (13%) units of fresh PRBCs and none of the old PRBCs were eligible for transfusion after 6 h of mechanical stress.
CONCLUSION:
When an austere combat environment was simulated for 24 h, fresh and old PRBC hemolysis rates were above the quality criteria. Currently, the technology to overcome this mechanical damage does not seem to exist. In light of the above data, a new national project is being performed.
24/10/2018
Don de sang: Capacité à l'exercice en baisse pour 2 Jours
The effect of a standard whole blood donation on oxygen uptake and exercise capacity: a systematic review and meta-analysis.
Van Remoortel H et Al. Transfusion. 2017 Feb;57(2):451-462
----------------------------------
----------------------------------
BACKGROUND:
Blood is a life-saving product for many people worldwide. Voluntary blood donation serves the demand for blood but there are concerns among potential donors about the impact of blood loss on exercise performance. This systematic review aimed to collect the best available evidence of the effect of a standard whole blood donation on aerobic exercise performance.
STUDY DESIGN AND METHODS:
Studies from six databases dealing with a standard whole blood donation (400-500 mL) followed by (sub)maximal exercise were retained. The outcomes included exercise-related blood variables (hemoglobin [Hb] concentration, hematocrit, and red blood cell count) and endurance exercise variables ((sub)maximal oxygen uptake, peak work rate, and time to exhaustion). Overall effects at different time points postdonation were investigated by performing meta-analyses and calculating mean differences (and 95% confidence intervals). The GRADE methodology (Grades of Recommendation, Assessment, Development, and Evaluation) was used to assess the quality of evidence.
RESULTS:
We identified 6237 references and finally included 18 before-after studies of low quality. Twenty-four to 48 hours after a blood donation, 1) Hb concentration was reduced (7% decrease) until 14 days after the blood donation (4% decrease), 2) maximal oxygen uptake(VO2max ) was lower (7% decrease), and 3) a reduction in maximal exercise capacity (10% decrease) was present.
CONCLUSION:
The best available evidence indicates that a standard whole blood donation (400-500 mL) leads to small but potentially physiologically important reductions in Hb levels, VO2max , and maximal exercise capacity in the first 2 days after the blood donation.
19/10/2018
Autotransfusion d'hémothorax et Plyo: Prudence
A small amount can make a difference: a prospective human study of the paradoxical coagulation characteristics of hemothorax.
The evacuated hemothorax has been poorly described because it varies with time, it has been found to be incoagulable, and its potential effect on the coagulation cascade during autotransfusion is largely unknown.
METHODS:
This is a prospective descriptive study of adult patients with traumatic chest injury necessitating tube thoracostomy. Pleural and venous samples were analyzed for coagulation, hematology, and electrolytes at 1 to 4 hours after drainage. Pleural samples were also analyzed for their effect on the coagulation cascade via mixing studies.
RESULTS:
Thirty-four subjects were enrolled with a traumatic hemothorax. The following measured coagulation factors were significantly depleted compared with venous blood: international normalized ratio (>9 vs 1.1) (P < .001) and activated partial thromboplastin time (aPTT) (>180 vs 24.5 seconds) (P < .001). Mixing studies showed a dose-dependent increase in coagulation dilutions through 1:8 (P < .05).
CONCLUSIONS:
An evacuated hemothorax does not vary in composition significantly with time and is incoagulable alone. Mixing studies with hemothorax plasma increased coagulation, raising safety concerns.
03/10/2018
Plaquettes et golden hour: Possible ?
Intravenous synthetic platelet (SynthoPlate) nanoconstructs reduce bleeding and improve 'golden hour' survival in a porcine model of traumatic arterial hemorrhage.
Traumatic non-compressible hemorrhage is a leading cause of civilian and military mortality and its treatment requires massive transfusion of blood components, especially platelets. However, in austere civilian and battlefield locations, access to platelets is highly challenging due to limited supply and portability, high risk of bacterial contamination and short shelf-life. To resolve this, we have developed an I.V.-administrable 'synthetic platelet' nanoconstruct (SynthoPlate), that can mimic and amplify body's natural hemostatic mechanisms specifically at the bleeding site while maintaining systemic safety.

Previously we have reported the detailed biochemical and hemostatic characterization of SynthoPlate in a non-trauma tail-bleeding model in mice. Building on this, here we sought to evaluate the hemostatic ability of SynthoPlate in emergency administration within the 'golden hour' following traumatic hemorrhagic injury in the femoral artery, in a pig model. We first characterized the storage stability and post-sterilization biofunctionality of SynthoPlate in vitro. The nanoconstructs were then I.V.-administered to pigs and their systemic safety and biodistribution were characterized. Subsequently we demonstrated that, following femoral artery injury, bolus administration of SynthoPlate could reduce blood loss, stabilize blood pressure and significantly improve survival. Our results indicate substantial promise of SynthoPlate as a viable platelet surrogate for emergency management of traumatic bleeding.
30/07/2018
Vitamine C pour la coagulopathie du traumatisé sévère
Interventional Vitamin C: A Strategy for Attenuation of Coagulopathy and Inflammation in a Swine Polytrauma Model
Reynolds PS et Al. J Trauma Acute Care Surg. 2018 DOI:10.1097/TA.0000000000001844
-----------------------------
L'emploi de vitamine C à haute dose est connue dans le cadre de la réanimation du brûlé (1). Elle limiterait la fuite capillaire du fait de propriétés anti-oxydantes. Les auteurs rapportent un intérêt expérimental modéré mais prometteur à leur avis.
-----------------------------
BACKGROUND
Coagulopathy and inflammation induced by hemorrhagic shock and traumatic injury are associated with increased mortality and morbidity. Vitamin C (VitC) is an antioxidant with potential protective effects on the pro-inflammatory and pro-coagulant pathways. We hypothesized that high-dose VitC administered as a supplement to fluid resuscitation would attenuate inflammation, coagulation dysfunction, and end-organ tissue damage in a swine model of polytrauma and hemorrhage.
METHODS
Male Sinclair swine (n = 24; mean body weight 27 kg) were anesthetized, intubated, mechanically ventilated, and instrumented for physiological monitoring. Following stabilization, swine were subjected to shock/traumatic injury (hypothermia, liver ischemia and reperfusion, comminuted femur fracture, hemorrhagic hypotension), resuscitated with 500mL of hydroxyethyl starch, and randomized to receive either intravenous saline (NS), low dose VitC (50mg/kg; LO), or high dose VitC (200 mg/kg; HI). Hemodynamics, blood chemistry, hematology, and coagulation function (ROTEM) were monitored to 4 hours post-resuscitation. Histological and molecular analyses were obtained for liver, kidney, and lung.
RESULTS
Compared to VitC animals, NS swine showed significant histological end-organ damage, elevated acute lung injury scores, and increased mRNA expression of tissue pro-inflammatory mediators (IL-1, IL-8, TNF), PAI-1, and TF. There were no statistically significant differences between treatment groups on MAP or univariate measures of coagulation function; however, NS showed impaired multivariate clotting function at 4 hours.
CONCLUSIONS
Although correction of coagulation dysfunction was modest, intravenous high-dose VitC may mitigate the pro-inflammatory/pro-coagulant response that contributes to multiple organ failure following acute severe polytrauma.
25/07/2018
Plasma et MEDEVAC, Obligation ? OUI
Prehospital plasma during air medical transport in trauma patients at risk for hemorrhagic shock.
----------------------------
Confirmation par l'étude PAMPER de tout l'intérêt de l'apport préhospitalier précoce d'au moins deux unités de plasma. Cette étude porte sur plus de 500 traumatisés graves essentiellement fermés, surtout secondaires à des AVP avec tout de même une motralité qui paraît très élevée. Il semblerait que l'administration avant l'arrivée en structure chirurgicale (mesurée dans ce travail à 1h) soit déterminante. Cependant, elle n'inclue que 20% de plaies pénétrantes sans faire mention d'IED, ce qui limite fortement une extrapolation simple aux blessés par arme de guerre., ce d'autant que le tableau 2 montre que dans ce sous groupe le traitement conventionnel ferait mieux en terme de survie. Un éditorial abordant les problématiques "logistiques" liée à l'emploi du plasma accompagne cette publication (1). Lire également (2, 3).
----------------------------
BACKGROUND:
After a person has been injured, prehospital administration of plasma in addition to the initiation of standard resuscitation procedures in the prehospital environment may reduce the risk of downstream complications from hemorrhage and shock. Data from large clinical trials are lacking to show either the efficacy or the risks associated with plasma transfusion in the prehospital setting.
METHODS:
To determine the efficacy and safety of prehospital administration of thawed plasma in injured patients who are at risk for hemorrhagic shock, we conducted a pragmatic, multicenter, cluster-randomized, phase 3 superiority trial that compared the administration of thawed plasma with standard-care resuscitation during air medical transport. The primary outcome was mortality at 30 days.
RESULTS:
A total of 501 patients were evaluated: 230 patients received plasma (plasma group) and 271 received standard-care resuscitation (standard-care group). Mortality at 30 days was significantly lower in the plasma group than in the standard-care group (23.2% vs. 33.0%; difference, -9.8 percentage points; 95% confidence interval, -18.6 to -1.0%; P=0.03). A similar treatment effect was observed across nine prespecified subgroups (heterogeneity chi-square test, 12.21; P=0.79). Kaplan-Meier curves showed an early separation of the two treatment groups that began 3 hours after randomization and persisted until 30 days after randomization (log-rank chi-square test, 5.70; P=0.02). The median prothrombin-time ratio was lower in the plasma group than in the standard-care group (1.2 [interquartile range, 1.1 to 1.4] vs. 1.3 [interquartile range, 1.1 to 1.6], P<0.001) after the patients' arrival at the trauma center. No significant differences between the two groups were noted with respect to multiorgan failure, acute lung injury-acute respiratory distress syndrome, nosocomial infections, or allergic or transfusion-related reactions.
CONCLUSIONS:
In injured patients at risk for hemorrhagic shock, the prehospital administration of thawed plasma was safe and resulted in lower 30-day mortality and a lower median prothrombin-time ratio than standard-care resuscitation.
| Tags : coagulopathie
08/07/2018
TXA et Plasma: A faire simultanément ?
BACKGROUND:
Traumatic coagulopathy has now been well characterized and carries high rates of mortality owing to bleeding. A 'factor-based' resuscitation strategy using procoagulant drugs and factor concentrates in lieu of plasma is being used by some, but with little evidentiary support. We sought to evaluate and compare resuscitation strategies using combinations of tranexamic acid (TXA), prothrombin complex concentrate (PCC), and fresh frozen plasma (FFP).
METHODS:
Sixty adult swine underwent 35% blood volume hemorrhage combined with a truncal ischemia-reperfusion injury to produce uniform shock and coagulopathy. Animals were randomized to control (n = 12), a single-agent group (TXA, n = 10; PCC, n = 8; or FFP, n = 6) or combination groups (TXA-FFP, n = 10; PCC-FFP, n = 8; TXA-PCC, n = 6). Resuscitation was continued to 6 hours. Key outcomes included hemodynamics, laboratory values, and rotational thromboelastometry. Results were compared between all groups, with additional comparisons between FFP and non-FFP groups.
RESULTS:
All 60 animals survived to 6 hours. Shock was seen in all animals, with hypotension (mean arterial pressure, 44 mm Hg), tachycardia (heart rate, 145), acidosis (pH 7.18; lactate, 11), anemia (hematocrit, 17), and coagulopathy (fibrinogen, 107). There were clear differences between groups for mean pH (p = 0.02), international normalized ratio (p < 0.01), clotting time (CT; p < 0.01), lactate (p = 0.01), creatinine (p < 0.01), and fibrinogen (p = 0.02). Fresh frozen plasma groups had significantly improved resuscitation and clotting parameters (Figures), with lower lactate at 6.5 versus 8.4 (p = 0.04), and increased fibrinogen at 126 versus 95 (p < 0.01). Rotational thromboelastometry also demonstrated shortened CT at 60 seconds in the FFP group vs 65 seconds in the non-FFP group (p = 0.04).
CONCLUSION:
When used to correct traumatic coagulopathy, combinations of FFP with TXA or PCC were superior in improving acidosis, coagulopathy, and CT than when these agents are given alone or in combination without plasma. Further validation of pure factor-based strategies is needed.
| Tags : hémorragie
11/05/2018
Plasma lyophilisé: Utile et mieux que PFC ?
French lyophilized plasma versus fresh frozen plasma for the initial management of trauma-induced coagulopathy: a randomized open-label trial
Garrigue D et AL; J Thromb Haemost. 2018 Mar;16(3):481-489
--------------------------
Le plasma lyophilisé retrouve d'évidence, en matière de réanimation transfusionnelle notamment celle du traumatisé, toute la place qu'il avait perdu dans les années 1980. Son atout majeur est sa disponibilité qui permet un apport bien plus précoce que le PFC. Ce travail en montre tout l'intérêt. On peut néanmoins dans ce document se poser la question de la similarité des groupes étudiés et des critères d'évaluation de la coagulopathie ?
--------------------------
Essentials
- An immediate supply of plasma in case of trauma-induced coagulopathy is required.
- The Traucc trial compared French Lyophilised Plasma (FLyP) and Fresh Frozen Plasma (FFP)
- FLyP achieved higher fibrinogen concentrations compared with FFP.
- FLyP led to a more rapid coagulopathy improvement than FFP.
Background
Guidelines recommend beginning hemostatic resuscitation immediately in trauma patients. We aimed to investigate if French lyophilized plasma (FLyP) was more effective than fresh frozen plasma (FFP) for the initial management of trauma-induced coagulopathy.
Methods In an open-label, phase 3, randomized trial (NCT02750150), we enrolled adult trauma patients requiring an emergency pack of 4 plasma units within 6 h of injury. We randomly assigned patients to receive 4-FLyP units or 4-FFP units. The primary endpoint was fibrinogen concentration at 45 min after randomization. Secondary outcomes included time to transfusion, changes in hemostatic parameters at different time-points, blood product requirements and 30-day in-hospital mortality.
Results
Forty-eight patients were randomized (FLyP, n = 24; FFP, n = 24). FLyP reduced the time from randomization to transfusion of first plasma unit compared with FFP (median[IQR],14[5-30] vs. 77[64-90] min). FLyP achieved a higher fibrinogen concentration 45 min after randomization compared with FFP (baseline-adjusted mean difference, 0.29 g L-1 ; 95% confidence interval [CI], 0.08-0.49) and a greater improvement in prothrombin time ratio, factor V and factor II. The between-group differences in coagulation parameters remained significant at 6 h. FLyP reduced fibrinogen concentrate requirements. Thirty-day in-hospital mortality rate was 22% with FLyP and 29% with FFP.
Conclusion FLyP led to a more rapid, pronounced and extended increase in fibrinogen concentrations and coagulopathy improvement compared with FFP in the initial management of trauma patients. FLyP represents an attractive option for trauma management, especially when facing logistical issues such as combat casualties or mass casualties related to terror attacks or disasters.
12/04/2018
Prélèvement de sang frais: VO2 Altérée
"Immediate effects of blood donation on physical and cognitive performance - A randomizedcontrolled double blinded trial"
The success of implementing Damage Control Resuscitation principles pre-hospital has been at the expense of several logistic burdens including the requirements for resupply, and the question of donor safety during the development of whole blood programs. Previous studies have reported effects on physical performance after blood donation, however none have investigated the effects of blooddonation on cognitive performance.
METHOD:
We describe a prospective double blinded, randomized controlled study comprised of a battery of tests: three cognitive tests, and VO2max testing on a cycle ergometer. Testing was performed 7 days before blinded donation (Baseline day), immediately after donation(Day 0), and 7 days (Day 7) after donation. The inclusion criteria included being active blood donors at the Haukeland University Hospital blood bank where eligibility requirements were met on the testing days and providing informed consent. Participants were randomized to either the experimental (n=26) or control group (n=31). Control group participants underwent a 'mock donation" in which a phlebotomy needle was placed but blood was not withdrawn.
RESULTS:
In the experimental group, mean VO2max declined 6% from 41.35 +/-1.7 /(min.kg) at baseline to 39.0 +/-1.6 /(min.kg) on Day 0, and increased to 40.51 +/-1.5 /(min.kg) on Day 7. Comparable values in the control group were 42.1 +/-1.8 /(min.kg) at baseline, 41.6 +/-1.8 /(min.kg) on Day 1 (1% decline from basline), and 41.8 +/-1.8 /(min.kg) on Day 7. Comparing scores of all three cognitive tests on Day 0 and Day 7 showed no significant differences, p>0.05.
CONCLUSION:
Our main findings are that executive cognitive and physical performance were well-maintained after whole blood donation in healthy blood donors. The findings inform post-donation guidance on when donors may be required to return to duty.
03/01/2018
Fibrinogène: En 1er chez les canadiens
Fibrinogen Concentrate in the Special Operations Forces Environment
Sanders S et al. Military Medecine, 00, 0/0:1, 2017
------------------------------------
Un choix raisonné et différent (Apport de 6 g de fibrinogène) des armées françaises qui s'intègre dans les nouvelles stratrégies transfusionnelles de prise en charge du blessé de guerre où la place de la transfusion de sang frais le fibrinogène et le plasma ont une place essentielle (1,2)
Aller sur le forum coagulopathie
------------------------------------
Introduction:
Hemorrhage is the most common cause of death among Special Operations Force (SOF) soldiers. Bringing remote damage control resuscitation into the far-forward combat environment is logistically challenging, as it requires blood products that generally require a robust cold chain. Alternatively, lyophilized products such as fibrinogen concentrate, which does not require thawing or blood group compatibility testing before use, might be advantageous in damage control resuscitation in the battlefield. In this report, we review the evidence for the use of fibrinogen concentrate in the Canadian SOF environment.
Materials and Methods:
The literature on the use of fibrinogen concentrate in the trauma setting was reviewed by Canadian Forces Services Working Group, in three separate meetings. Multiple stakeholders were consulted to obtain authoritative perspectives from subject matter experts on the use of fibrinogen concentrate in the Canadian SOF environment.

We also conducted a comparison review of fibrinogen content, pathogen risk, shelf life, and methods required for use for fresh frozen plasma, cryoprecipitate, and fibrinogen concentrate relevant to their application in the far-forward combat environment.
Results:
Indications and a protocol for the use of fibrinogen as an adjunct to fresh whole blood were formulated based on a literature review and clinical expert opinion. Alternative strategies and other lyophilized blood products were considered before selecting fibrinogen concentrate as the lyophilized blood product of choice. Fibrinogen concentrate is an ABO-universal blood product with an excellent safety profile. Training was conducted by subject matter experts within civilian trauma centers and at military training facilities. The clinical efficacy and safety were confirmed by monitoring the use of fibrinogen concentrate in deployed combat settings.
Conclusion:
Fibrinogen concentrate is a useful adjunct to remote damage control resuscitation in the SOF environment. Fibrinogen concentrate was found to be robust for transport into the SOF environment and is widely accepted among SOF operators and medics
24/12/2017
AAST 2017: Utilité du TXA encore questionnée
Late TXA utilization is associated with Iincreased blood product transfusion: A secondary analysisof the pragmatic randomized optimal platelet and plasma ratios (PROPPR) study
Aravind K. et AL . Session: XIIIA: Papers 45-54: Preclincial/Translational Science Paper 52
-----------------------------------
Ce poster présenté au dernier AAST 2017 est une ré-interprétation de données de l'étude POPR. Il semblerait qu'au delà des premières heures l'administration précoce (dans la 1ère heure) de TXA n'améliore pas la survie à 24h et un mois. L'administration tardive (après la 1ère heure mais avant la 3ème) majorerait les besoins transfusionnels. Les auteurs de ce poster attirent l'attention sur la disparité des groupes comparés, ce qui peut expliquer leurs résultats.
-----------------------------------
Introduction:
Exsanguination is the leading cause of preventable death after trauma. In addition to a balanced ratio blood component strategy, tranexamic acid (TXA) is used as an adjunct in hemorrhaging patients. This secondary analysis was performed to determine the incidence of TXA utilization and outcome in patients predicted to receive a massive transfusion (MT) in level 1 trauma centers.
Methods:
Trauma patients who were predicted to require a MT and admitted to 12 level I North American trauma centers were studied. Patients were divided into those who received TXA and those who did not. We examined 3 hour, 24 hour, and 30 day mortality. We also examined incidence of thromboembolic events, blood product administration within the first 24 hours, length of stay (hospital free days), ICU free days, as well as development of complications including acute respiratory distress syndrome (ARDS), acute kidney injury (AKI), sepsis, and multisystem organ failure (MOF). In our multivariate analysis, we controlled for Injury Severity Score (ISS), Glasgow Coma Scale (GCS), treatment group, mechanism of injury, hypotension and/or tachycardia on admission, geriatric patient (age > 65), and site as independent variables.
Results:
137 out of 680 (20.1%) patients in the PROPPR study received TXA with 130 patients receiving TXA within the first 3 hours after admission. Other adjunctive therapies administered included cryoprecipitate (25.4%), and others (6.6%). The incidence of TXA administration did not differ between the ratio groups (50.3% vs 47.4%, p=0.55), but patients receiving TXA were more severely injured with a median ISS of 34(21) vs 26(20), p<0.01 and a lower median(IQR) GCS of 9(12) vs 14(12), p<0.01. Multivariate linear regression analysis revealed no association between TXA administration and blood transfusion requirements (Table 1). Further analysis revealed that patients who received late (from >1 hour to ≤3 hours after arrival) TXA (41 patients) experienced increased blood requirements in the first 24 hours (Table 1) compared to those remaining 543 patients that did not receive TXA. There was no difference in blood product requirement in those patients who received TXA early (≤1 hour) (89 patients) versus those that did not receive TXA. In patients that receiv ed TXA, there was an increased incidence of ARDS(OR (95% CI) 1.99 (1.06,3.73), p=0.03), AKI (1.90 (1.13,3.20), p=0.01), and MOF (4.18 (1.52,11.48), p<0.01) even when controlling for the factors mentioned above. There was also a difference in adjusted 3 hour mortality (OR (95% CI) 0.22 (0.07,0.73), p=0.01) but not 24 hour (0.61 (0.30,1.24), p=0.18) or 30 day mortality (1.42 (0.78,2.59), p=0.25) for any TXA administration. There was no difference in adjusted thromboembolic events or adjusted length of stay. Subgroup analysis with additional variables that were found to have a difference between groups with a p<0.20 were added to the regression model including hematocrit, platelet count, international normalized ratio, creatinine, lactate, and R value on thrombelastography. This analysis showed an increase in PRBC transfusion with late TXA administration (16 out of 232 patients, 7.51 (0.46,14.56), p=0.04), but showed no difference in FFP or platelet administration.
Conclusion: Early TXA use was not associated with improved outcomes. Late TXA use was associated with increased blood product resuscitation. TXA administration in general was associated with improved 3 hour mortality. This did not translate to an improvement in mortality at 24 hours or 30 days. There was a significant increase in the incidence of ARDS, AKI, and MOF in patients who received TXA but this analysis is limited by the differences in the 2 populations despite attempts to control for them
23/12/2017
Le glycoalyx: Une cible thérapeutique
-----------------------------------
La correction précoce de la coagulopathie traumatique est un des objectifs fondamentaux de la prise en charge du traumatisé sévère et en particulier des blessés de guerre. L'emploi de plasma lyophilisé / fibrinogène / fractions coagulantes concentrées et la transfusion de sang frais se fait selon des stratégies diverses visant à compenser les déficits observés (1) . La restauration d'une perméabilité capillaire est également une voie possible. On peut considérer que l'efficacité du plasma est en +/- grande partie liée à sa capacité à restaurer le glycocalyx endothélial (2). Le travail présenté est en faveur de l'emploi, à l'instar de l'acide tranexaminique pour la fibrinolyse, d'ajuvants pharmacologiques aux solutés de remplissage pour limiter l'atteinte du glycoclayx.
Allez sur le forum coagulopathie/transfusion
Mieux comprende le concept de perméabilité capillaire
-----------------------------------
INTRODUCTION:
There is interest in the small-volume therapeutic use of adjunct drugs for treating hemorrhagic shock (HS). However, critical information is only partially available on mechanisms of action of promising compounds such as adenosine-lidocaine-magnesium (ALM), beta-hydroxybutyrate plus melatonin (BHB/M), and poloxamer 188 (P-188). Therefore, we tested the hypothesis that these adjuncts would reverse HS-induced damage to microvascular endothelial glycocalyx and hemodynamics.
METHODS:
After baseline, 40% of total blood volume was removed from 44 anesthetized Sprague-Dawley male rats. One hour after hemorrhage, animals were resuscitated using ALM, BHB/M, or P-188 followed by lactated Ringer's (LR, 15 mL/kg). Control animals were not treated (SHAM) or received LR alone. Sampled blood was used to quantify shed syndecan-1 in plasma; multiple systemic physiological parameters were recorded. In vivo glycocalyx thickness, microvascular permeability, and microhemodynamics were evaluated in >200 cremaster venules using intravital videomicroscopy.
RESULTS:
Compared with baseline, resuscitation using adjuncts was associated with glycocalyx restoration of 97 ± 9% (ALM), 75 ± 8% (BHB/M), and 85 ± 5% (P-188): significantly higher than LR-only (56 ± 4%). Significantly better permeability, similar to SHAM values, was measured after ALM and P-188, and low plasma syndecan-1 levels were measured after resuscitation with all adjuncts. Microhemodynamic changes were relatively small while systemic parameters such as mean arterial pressure and lactate improved but remained below or above the baseline, respectively, as expected from this hypotensive resuscitation model.
CONCLUSION:
The drugs ALM, BHB/M, and P-188 provide beneficial effects as adjuncts to hypotensive resuscitation in this HS model by mechanisms involving changes at the microvascular level including the glycocalyx.
| Tags : coagulopathie, transfusion
21/12/2017
Pour du sang à l'avant: Le drone !
L'emploi du sang se heurte à de nombreux problèmes. Le recours à des drones n'est pas une utopie. Tout ceci est très étudié par les pays dont le maillage hospitaliers et les infrastructures sont peu développés.
Les drones font partie du futur (1) et leur autonomie est un atout précieux pour qu’ils puissent devenir un moyen de porter rapidement secours aux individus. Ainsi, les aéronefs sont dès à présent envisagés comme une alternative pour livrer des équipements médicaux dans des lieux qui pourraient s’avérer plus compliqués à traverser pour l’être humain (2)
« Les drones pourraient être le meilleur système de livraison des équipements médicaux du vingt-et-unième siècle "
Diverses technologies de drone peuvent être employées. Un modèle tanzanien parait bien opérant (3) et commence à être utilisé en routine au rwanda pour la délivrance de sang (4) . Certains ont démontré leur capacité à oeuvrer dans un rayon de 260 km (5, lire cette publication US).
| Tags : drone
02/12/2017
Fibrinogène en PREMIER pour le trauma
BACKGROUND:
Effective treatment of trauma-induced coagulopathy is important; however, the optimal therapy is still not known. We aimed to compare the efficacy of first-line therapy using fresh frozen plasma (FFP) or coagulation factor concentrates (CFC) for the reversal of trauma-induced coagulopathy, the arising transfusion requirements, and consequently the development of multiple organ failure.
METHODS:
This single-centre, parallel-group, open-label, randomised trial was done at the Level 1 Trauma Center in Innsbruck Medical University Hospital (Innsbruck, Austria). Patients with trauma aged 18-80 years, with an Injury Severity Score (ISS) greater than 15, bleeding signs, and plasmatic coagulopathy identified by abnormal fibrin polymerisation or prolonged coagulation time using rotational thromboelastometry (ROTEM) were eligible. Patients with injuries that were judged incompatible with survival, cardiopulmonary resuscitation on the scene, isolated brain injury, burn injury, avalanche injury, or prehospital coagulation therapy other than tranexamic acid were excluded. We used a computer-generated randomisation list, stratification for brain injury and ISS, and closed opaque envelopes to randomly allocate patients to treatment with FFP (15 mL/kg of bodyweight) or CFC (primarily fibrinogen concentrate [50 mg/kg of bodyweight]). Bleeding management began immediately after randomisation and continued until 24 h after admission to the intensive care unit. The primary clinical endpoint was multiple organ failure in the modified intention-to-treat population (excluding patients who discontinued treatment). Reversal of coagulopathy and need for massive transfusions were important secondary efficacy endpoints that were the reason for deciding the continuation or termination of the trial. This trial is registered with ClinicalTrials.gov, number NCT01545635.
FINDINGS:
Between March 3, 2012, and Feb 20, 2016, 100 out of 292 screened patients were included and randomly allocated to FFP (n=48) and CFC (n=52). Six patients (four in the FFP group and two in the CFC group) discontinued treatment because of overlooked exclusion criteria or a major protocol deviation with loss of follow-up. 44 patients in the FFP group and 50 patients in the CFC group were included in the final interim analysis.
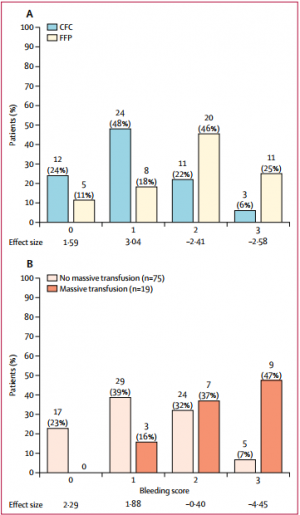
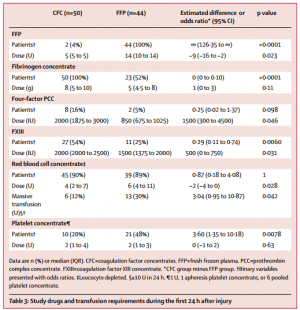
The study was terminated early for futility and safety reasons because of the high proportion of patients in the FFP group who required rescue therapy compared with those in the CFC group (23 [52%] in the FFP group vs two [4%] in the CFC group; odds ratio [OR] 25·34 [95% CI 5·47-240·03], p<0·0001) and increased needed for massive transfusion (13 [30%] in the FFP group vs six [12%] in the CFC group; OR 3·04 [0·95-10·87], p=0·042) in the FFP group. Multiple organ failure occurred in 29 (66%) patients in the FFP group and in 25 (50%) patients in the CFC group (OR 1·92 [95% CI 0·78-4·86], p=0·15).
INTERPRETATION:
Our results underline the importance of early and effective fibrinogen supplementation for severe clotting failure in multiple trauma. The available sample size in our study appears sufficient to make some conclusions that first-line CFC is superior to FFP.
01/12/2017
Transfusion à l'avant: Surtout Plyo, et le sang ?
Early transfusion on battlefield before admission to role 2: A preliminary observational study during “Barkhane” operation in Sahel
Vitalys V. et Al. https://doi.org/10.1016/j.injury.2017.11.029
---------------------------------------
Ce document rapporte essentiellement l'emploi de PLyo chez des blessés de guerre avant leur prise en charge en role 2. Beaucoup est écrit sur la faisabilité d'une telle pratique. On rappelle que l'emploi du plasma lyophilisé n'est pas une nouveauté y compris en milieu précaire (1). Le plasma lyophilisé était d'emploi très courant dans les années 70 et début 80, à un tel point qu'il était pratiquement utilisé comme soluté de remplissage chez le traumatisé routier grave. Il s'agit donc d'une redécouverte et on n'est pas surpris que ceci puisse se retrouver 30 ans après avec un produit notoirement sécurisé (2).
Il est dommage que l'article ne détaille pas plus cet emploi au niveau des role 1 qui est le lieu de début de transfusion pour 5 des blessés transfusés sur 7, ce qui n'est pas vraiment mis en avant dans ce travail. La reconstitution du Plyo est simple, mais probablement moins aisée dans une voilure tournante que dans un Casanurse ou au sol. Cet article met également en avant l'absence de transfusion de sang total, ce qui est étonnant pour une pratique rentrée dans les moeurs chez nos collègues US et UK. Enfin on retrouve l'emploi de fibrinogène, dont l'apport suffisant ne peut être assuré par le PLyo seul, et que certains proposent en première ligne de fractions coagulantes (3). Chose intéressante deux échecs de reconstitution du Plyo ont été observées.
Un article très intéressant, dont plusieurs auteurs sont en charge du développement du PLyo, avec dans la partie discussion un bon rappel des problématiques qui se posent dans de telles circonstances. Il est dommage que le regroupement des lésions du bassin avec l'atteinte des membres ait été fait car les problématiques de contrôle des hémorragies sont différentes. De même la description des lésions observées dans le groupe transfusé (qui ne sont pas tous porteurs d'un garrot), et l'analyse spécifique du groupe présentant une hémorragie encore active aurait été méritées.
---------------------------------------
Introduction
Haemorrage is the leading cause of death after combat related injuries and bleeding management is the cornerstone of management of these casualties. French armed forces are deployed in Barkhane operatio n in the Sahel-Saharan Strip who represents an immense area. Since this constraint implies evacuation times beyond doctrinal timelines, an institutional decision has been made to deploy blood products on the battlefield and transfuse casualties before role 2 admission if indicated. The purpose of this study was to evaluate the transfusion practices on battlefield during the first year following the implementation of this policy.
Materials and methods
prospective collection of data about combat related casualties categorized alpha evacuated to a role 2. Battlefield transfusion was defined as any transfusion of blood product (red blood cells, plasma, whole blood) performed by role 1 or Medevac team before admission at a role 2. Patients’ characteristics, battlefield transfusions’ characteristics and complications were analysed.
Results
During the one year study, a total of 29 alpha casualties were included during the period study. Twenty-eight could be analysed, 7/28 (25%) being transfused on battlefield, representing a total of 22 transfusion episodes. The most frequently blood product transfused was French lyophilized plasma (FLYP). Most of transfusion episodes occurred during medevac. Compared to non-battlefield transfused casualties, battlefield transfused casualties suffered more wounded anatomical regions (median number of 3 versus 2, p = 0.04), had a higher injury severity score (median ISS of 45 versus 25, p = 0,01) and were more often transfused at role 2, received more plasma units and whole blood units. There was no difference in evacuation time to role 2 between patients transfused on battlefield and non-transfused patients. There was no complication related to battlefield transfusions. Blood products transfusion onset on battlefield ranged from 75 min to 192 min after injury.
Conclusion
Battlefield transfusion for combat-related casualties is a logistical challenge. Our study showed that such a program is feasible even in an extended area as Sahel-Saharan Strip operation theatre and reduces time to first blood product transfusion for alpha casualties. FLYP is the first line blood product on the battlefield
| Tags : transfusion
31/08/2017
TXA: Peut être pas si évident
Military use of TXA in combat trauma: Does it matter?
BACKGROUND:
Tranexamic acid (TXA) has been previously reported to have a mortality benefit in civilian and combat-related trauma, and was thus added to the Joint Theater Trauma System Damage Control Resuscitation Clinical Practice Guideline. As part of ongoing system-wide performance improvement, the use of TXA has been closely monitored. The goal was to evaluate the efficacy and safety of TXA use in military casualties and provide additional guidance for continued use.
METHODS:
A total of 3,773 casualties were included in this retrospective, observational study of data gathered from a trauma registry. The total sample, along with 3 sub-samples for massive transfusion patients (n=784), propensity-matched sample (n=1,030) and US/NATO military (n=1,262), were assessed for administration of TXA and time from injury to administration of TXA. Outcomes included mortality and occurrence of pulmonary embolism (PE) and deep vein thrombosis (DVT). Multivariable proportional hazards regression models with robust standard error estimates were used to estimate hazard ratios (HR) for assessment of outcomes while controlling for covariates.
RESULTS:
Results of univariate and multivariate analyses of the total sample (HR=0.97; 95%CI 0.62-1.53; p=0.86); massive transfusion sample (HR=0.84; 95%CI 0.46-1.56; p=0.51); propensity-matched sample (HR=0.68; 95%CI 0.27-1.73; p=0.34); and US/NATO military sample (HR=0.76; 95%CI 0.30-1.92; p=0.48) indicate no statistically significant association between TXA use and mortality. Use of TXA was associated with increased risk of PE in the total sample (HR=2.82; 95%CI 2.08-3.81; p<0.001); massive transfusion sample (HR=3.64; 95%CI 1.96-6.78; p=0.003); US/NATO military sample (HR=2.55; 95%CI 1.73-3.69; p=0.002); but not the propensity-matched sample (HR=3.36; 95%CI 0.80-14.10; p=0.10). TXA was also associated with increased risk of DVT in the total sample (HR=2.00; 95%CI 1.21-3.30; p=0.02) and US/NATO military sample (HR=2.18; 95%CI 1.20-3.96; p=0.02).
CONCLUSIONS:
In the largest study on TXA use in a combat trauma population, TXA was not significantly associated with mortality, due to lack of statistical power. However, our HR estimates for mortality among patients who received TXA are consistent with previous findings from the CRASH2 trial. At the same time, continued scrutiny and surveillance of TXA use in military trauma, specifically for prevention of thromboembolic events, is warranted.
| Tags : transfusion
