30/12/2018
HEXACYL: Oui , mais attention à la MTE
Tranexamic acid administration is associated with an increased risk of posttraumatic venous thromboembolism.
BACKGROUND:
Tranexamic acid (TXA) is used as a hemostatic adjunct for hemorrhage control in the injured patient and reduces early preventable death. However, the risk of venous thromboembolism (VTE) has been incompletely explored. Previous studies investigating the effect of TXA on VTE vary in their findings. We performed a propensity matched analysis to investigate the association between TXA and VTE following trauma, hypothesizing that TXA is an independent risk factor for VTE.
METHODS:
This retrospective study queried trauma patients presenting to a single Level I trauma center from 2012 to 2016. Our primary outcome was composite pulmonary embolism or deep vein thrombosis. Mortality, transfusion, intensive care unit and hospital lengths of stay were secondary outcomes. Propensity matched mixed effects multivariate logistic regression was used to determine adjusted odds ratio (aOR) and 95% confidence intervals (95% CI) of TXA on outcomes of interest, adjusting for prespecified confounders. Competing risks regression assessed subdistribution hazard ratio of VTE after accounting for mortality.
RESULTS:
Of 21,931 patients, 189 pairs were well matched across propensity score variables (standardized differences <0.2). Median Injury Severity Score was 19 (interquartile range, 12-27) and 14 (interquartile range, 8-22) in TXA and non-TXA groups, respectively (p = 0.19). Tranexamic acid was associated with more than threefold increase in the odds of VTE (aOR, 3.3; 95% CI, 1.3-9.1; p = 0.02).
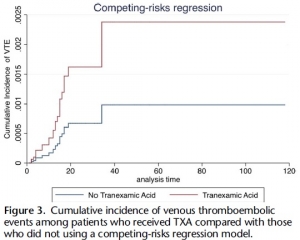
Tranexamic acid was not significantly associated with survival (aOR, 0.86; 95% CI, 0.23-3.25; p = 0.83). Risk of VTE remained elevated in the TXA cohort despite accounting for mortality (subdistribution hazard ratio, 2.42; 95% CI, 1.11-5.29; p = 0.03).
CONCLUSION:
Tranexamic acid may be an independent risk factor for VTE. Future investigation is needed to identify which patients benefit most from TXA, especially given the risks of this intervention to allow a more individualized treatment approach that maximizes benefits and mitigates potential harms.
| Tags : coagulopathie
25/07/2018
Plasma et MEDEVAC, Obligation ? OUI
Prehospital plasma during air medical transport in trauma patients at risk for hemorrhagic shock.
----------------------------
Confirmation par l'étude PAMPER de tout l'intérêt de l'apport préhospitalier précoce d'au moins deux unités de plasma. Cette étude porte sur plus de 500 traumatisés graves essentiellement fermés, surtout secondaires à des AVP avec tout de même une motralité qui paraît très élevée. Il semblerait que l'administration avant l'arrivée en structure chirurgicale (mesurée dans ce travail à 1h) soit déterminante. Cependant, elle n'inclue que 20% de plaies pénétrantes sans faire mention d'IED, ce qui limite fortement une extrapolation simple aux blessés par arme de guerre., ce d'autant que le tableau 2 montre que dans ce sous groupe le traitement conventionnel ferait mieux en terme de survie. Un éditorial abordant les problématiques "logistiques" liée à l'emploi du plasma accompagne cette publication (1). Lire également (2, 3).
----------------------------
BACKGROUND:
After a person has been injured, prehospital administration of plasma in addition to the initiation of standard resuscitation procedures in the prehospital environment may reduce the risk of downstream complications from hemorrhage and shock. Data from large clinical trials are lacking to show either the efficacy or the risks associated with plasma transfusion in the prehospital setting.
METHODS:
To determine the efficacy and safety of prehospital administration of thawed plasma in injured patients who are at risk for hemorrhagic shock, we conducted a pragmatic, multicenter, cluster-randomized, phase 3 superiority trial that compared the administration of thawed plasma with standard-care resuscitation during air medical transport. The primary outcome was mortality at 30 days.
RESULTS:
A total of 501 patients were evaluated: 230 patients received plasma (plasma group) and 271 received standard-care resuscitation (standard-care group). Mortality at 30 days was significantly lower in the plasma group than in the standard-care group (23.2% vs. 33.0%; difference, -9.8 percentage points; 95% confidence interval, -18.6 to -1.0%; P=0.03). A similar treatment effect was observed across nine prespecified subgroups (heterogeneity chi-square test, 12.21; P=0.79). Kaplan-Meier curves showed an early separation of the two treatment groups that began 3 hours after randomization and persisted until 30 days after randomization (log-rank chi-square test, 5.70; P=0.02). The median prothrombin-time ratio was lower in the plasma group than in the standard-care group (1.2 [interquartile range, 1.1 to 1.4] vs. 1.3 [interquartile range, 1.1 to 1.6], P<0.001) after the patients' arrival at the trauma center. No significant differences between the two groups were noted with respect to multiorgan failure, acute lung injury-acute respiratory distress syndrome, nosocomial infections, or allergic or transfusion-related reactions.
CONCLUSIONS:
In injured patients at risk for hemorrhagic shock, the prehospital administration of thawed plasma was safe and resulted in lower 30-day mortality and a lower median prothrombin-time ratio than standard-care resuscitation.
| Tags : coagulopathie
23/12/2017
Le glycoalyx: Une cible thérapeutique
-----------------------------------
La correction précoce de la coagulopathie traumatique est un des objectifs fondamentaux de la prise en charge du traumatisé sévère et en particulier des blessés de guerre. L'emploi de plasma lyophilisé / fibrinogène / fractions coagulantes concentrées et la transfusion de sang frais se fait selon des stratégies diverses visant à compenser les déficits observés (1) . La restauration d'une perméabilité capillaire est également une voie possible. On peut considérer que l'efficacité du plasma est en +/- grande partie liée à sa capacité à restaurer le glycocalyx endothélial (2). Le travail présenté est en faveur de l'emploi, à l'instar de l'acide tranexaminique pour la fibrinolyse, d'ajuvants pharmacologiques aux solutés de remplissage pour limiter l'atteinte du glycoclayx.
Allez sur le forum coagulopathie/transfusion
Mieux comprende le concept de perméabilité capillaire
-----------------------------------
INTRODUCTION:
There is interest in the small-volume therapeutic use of adjunct drugs for treating hemorrhagic shock (HS). However, critical information is only partially available on mechanisms of action of promising compounds such as adenosine-lidocaine-magnesium (ALM), beta-hydroxybutyrate plus melatonin (BHB/M), and poloxamer 188 (P-188). Therefore, we tested the hypothesis that these adjuncts would reverse HS-induced damage to microvascular endothelial glycocalyx and hemodynamics.
METHODS:
After baseline, 40% of total blood volume was removed from 44 anesthetized Sprague-Dawley male rats. One hour after hemorrhage, animals were resuscitated using ALM, BHB/M, or P-188 followed by lactated Ringer's (LR, 15 mL/kg). Control animals were not treated (SHAM) or received LR alone. Sampled blood was used to quantify shed syndecan-1 in plasma; multiple systemic physiological parameters were recorded. In vivo glycocalyx thickness, microvascular permeability, and microhemodynamics were evaluated in >200 cremaster venules using intravital videomicroscopy.
RESULTS:
Compared with baseline, resuscitation using adjuncts was associated with glycocalyx restoration of 97 ± 9% (ALM), 75 ± 8% (BHB/M), and 85 ± 5% (P-188): significantly higher than LR-only (56 ± 4%). Significantly better permeability, similar to SHAM values, was measured after ALM and P-188, and low plasma syndecan-1 levels were measured after resuscitation with all adjuncts. Microhemodynamic changes were relatively small while systemic parameters such as mean arterial pressure and lactate improved but remained below or above the baseline, respectively, as expected from this hypotensive resuscitation model.
CONCLUSION:
The drugs ALM, BHB/M, and P-188 provide beneficial effects as adjuncts to hypotensive resuscitation in this HS model by mechanisms involving changes at the microvascular level including the glycocalyx.
| Tags : coagulopathie, transfusion
29/07/2017
Fibrinogène ET Plasma
Relative effects of plasma, fibrinogen concentrate, and factor XIII on ROTEM coagulation profiles in an in vitro model of massive transfusion in trauma.
----------------------------------
Un travail qui est en faveur de l'association de fibrinogène et de plasma pour la prise en charge des hémorragies massives. Dans le contexte militaire, une stratégie basée sur l'apport initial du fibrinogène puis du Plyo apparaît censé (1). Lire les reco européennes.
----------------------------------
Massive traumatic haemorrhage is aggravated through the development of trauma-induced coagulopathy, which is managed by plasma transfusion and/or fibrinogen concentrate administration. It is yet unclear whether these treatments are equally potent in ensuring adequate haemostasis, and whether additional factor XIII (FXIII) administration provides further benefits.
In this study, we compared ROTEM whole blood coagulation profiles after experimental massive transfusion with different transfusion regimens in an in vitro model of dilution- and transfusion-related coagulopathy. Healthy donor blood was mixed 1 + 1 with six different transfusion regimens. Each regimen contained RBC, platelet concentrate, and either fresh frozen plasma (FFP) or Ringer's acetate (RA). The regimens were further augmented through addition of a low- or medium-dose fibrinogen concentrate and FXIII.
Transfusion with FFP alone was insufficient to maintain tissue-factor activated clot strength, coincidental with a deficiency in fibrin-based clot strength. Fibrinogen concentrate conserved, but did not improve coagulation kinetics and overall clot strength. Only combination therapy with FFP and low-dose fibrinogen concentrate improved both coagulation kinetics and fibrin-based clot strength. Administration of FXIII did not result in an improvement of clot strength. In conclusion, combination therapy with both FFP and low-dose fibrinogen concentrate improved clotting time and produced firm clots, representing a possible preferred first-line regimen to manage trauma-induced coagulopathy when RBC and platelets are also transfused. Further research is required to identify optimal first-line transfusion fluids for massive traumatic haemorrhage.
| Tags : coagulopathie
05/05/2017
Fractions coagulantes: Mieux que PFC ?
Reversal of trauma-induced coagulopathy using first-line coagulation factor concentrates or fresh frozen plasma (RETIC): a single-centre, parallel-group, open-label, randomised trial.
-------------------------------------------------
Un travail dont les conclusions sont très claires. La prise en charge de la coagulopathie traumatique bénéficie bien plus d'une stratégie d'apports ciblés de fractions coagulantes concentrées, notamment de fibrinogène ici 50 mg/kg, qu'une stratégie classique basée sur l'apport de PFC.
-------------------------------------------------
Background
Effective treatment of trauma-induced coagulopathy is important; however, the optimal therapy is still not known. We aimed to compare the efficacy of first-line therapy using fresh frozen plasma (FFP) or coagulation factor concentrates (CFC) for the reversal of trauma-induced coagulopathy, the arising transfusion requirements, and consequently the development of multiple organ failure.
Methods
This single-centre, parallel-group, open-label, randomised trial was done at the Level 1 Trauma Center in Innsbruck Medical University Hospital (Innsbruck, Austria). Patients with trauma aged 18–80 years, with an Injury Severity Score (ISS) greater than 15, bleeding signs, and plasmatic coagulopathy identified by abnormal fibrin polymerisation or prolonged coagulation time using rotational thromboelastometry (ROTEM) were eligible. Patients with injuries that were judged incompatible with survival, cardiopulmonary resuscitation on the scene, isolated brain injury, burn injury, avalanche injury, or prehospital coagulation therapy other than tranexamic acid were excluded. We used a computer-generated randomisation list, stratification for brain injury and ISS, and closed opaque envelopes to randomly allocate patients to treatment with FFP (15 mL/kg of bodyweight) or CFC (primarily fibrinogen concentrate [50 mg/kg of bodyweight]). Bleeding management began immediately after randomisation and continued until 24 h after admission to the intensive care unit. The primary clinical endpoint was multiple organ failure in the modified intention-to-treat population (excluding patients who discontinued treatment). Reversal of coagulopathy and need for massive transfusions were important secondary efficacy endpoints that were the reason for deciding the continuation or termination of the trial. This trial is registered with ClinicalTrials.gov, number NCT01545635.
Findings
Between March 3, 2012, and Feb 20, 2016, 100 out of 292 screened patients were included and randomly allocated to FFP (n=48) and CFC (n=52). Six patients (four in the FFP group and two in the CFC group) discontinued treatment because of overlooked exclusion criteria or a major protocol deviation with loss of follow-up. 44 patients in the FFP group and 50 patients in the CFC group were included in the final interim analysis. The study was terminated early for futility and safety reasons because of the high proportion of patients in the FFP group who required rescue therapy compared with those in the CFC group (23 [52%] in the FFP group vs two [4%] in the CFC group; odds ratio [OR] 25·34 [95% CI 5·47–240·03], p<0·0001) and increased needed for massive transfusion (13 [30%] in the FFP group vs six [12%] in the CFC group; OR 3·04 [0·95–10·87], p=0·042) in the FFP group. Multiple organ failure occurred in 29 (66%) patients in the FFP group and in 25 (50%) patients in the CFC group (OR 1·92 [95% CI 0·78–4·86], p=0·15).
Interpretation
Our results underline the importance of early and effective fibrinogen supplementation for severe clotting failure in multiple trauma. The available sample size in our study appears sufficient to make some conclusions that first-line CFC is superior to FFP.
| Tags : coagulopathie
02/02/2017
Coagulopathie: Une affaire personnelle
Individual clotting factor contributions to mortality following trauma.
-------------------------------
Un travail intéressant qui portant sur 1463 traumatisés d'ISS médian de 16 et qui met en avant l'existence de deux profils de coagulopathie traumatique (16% des patients pris en charge). Dans 49,30 des cas, un premier est en rapport avec des anomalies des facteurs II,VII,IX,X et protéine C . Le second profil (17% des patients) exprime des anomalies sur les facteurs V et VIII. Seuls seraient liés à une motralité à 28j. LA déplétion en facteur V serait associée à une mortalité à long terme. Ce travail milite pour une approche personnalisée de la ocaguklopathie traumatique.
-------------------------------
BACKGROUND:
Acute traumatic coagulopathy affects 20% to 30% of trauma patients, but the extensive collinearity of the coagulation cascade complicates attempts to clarify global clotting factor dysfunction. This study aimed to characterize phenotypes of clotting factor dysfunction and their contributions to mortality after major trauma.
METHODS:
This prospective cohort study examines all adult trauma patients of the highest activation level presenting to San Francisco General Hospital between February 2005 and February 2015. Factors II, V, VII, VIII, IX, and X and protein C activity on admission and mortality status at 28 days were assessed. Predictors of 28-day mortality in univariate analysis were included in multiple logistic regression controlling for traumatic brain injury (TBI), acidosis, age, and mechanism of injury. Principal component analysis was utilized to identify phenotypic coagulation.
RESULTS:
Complete coagulation factor data were available for 876 (61%) of 1,429 patients. In multiple logistic regression, factors V (odds ratio [OR], 0.86; 95% confidence interval [CI], 0.76-0.97), VIII (OR, 0.97; 95% CI, 0.95-0.99), and X (OR, 0.79; 95% CI, 0.68-0.92) and protein C (OR, 1.17; 95% CI, 1.05-1.30) significantly predicted 28-day mortality after controlling for age, base deficit, mechanism of injury, and TBI. Principal component analysis identified two significant principal components (Phenotypes 1 and 2) that accounted for 66.3% of the total variance. Phenotype 1 (factors II, VII, IX, and X and protein C abnormalities) explained 49.3% and was associated with increased injury, coagulopathy, TBI, and mortality. Phenotype 2 (factors V and VIII abnormalities) explained 17.0% and was associated with increased coagulopathy, blunt injury, and mortality. Only Phenotype 2 remained significantly associated with 28-day mortality in multiple logistic regression.
CONCLUSIONS:
Principal component analysis identified two distinct phenotypes within the entirety of global clotting factor abnormalities, and these findings substantiate the crucial association of factors V and VIII on mortality following trauma. This may be the first step toward identifying unique phenotypes after injury and personalizing hemostatic resuscitation.
| Tags : hémorragie, coagulopathie
21/01/2017
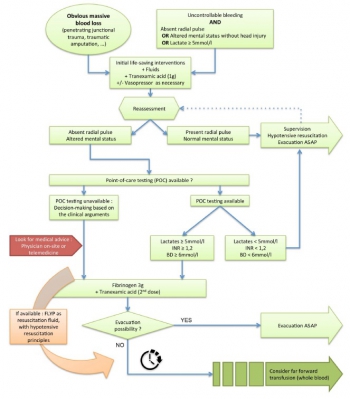
Coagulopathie: Fibrinogène avant PLyo, mais les 2 et + tôt ?
Despite the early uses of tourniquets and haemostatic dressings, blood loss still accounts for the vast majority of preventable deaths on the battlefield. Over the last few years, progress has been made in the management of such injuries, especially with the use of damage control resuscitation concepts. The early application of these procedures, on the field, may constitute the best opportunity to improve survival from combat injury during remote operations.
DATA SOURCES:
Currently available literature relating to trauma-induced coagulopathy treatment and far-forward transfusion was identified by searches of electronic databases. The level of evidence and methodology of the research were reviewed for each article. The appropriateness for field utilisation of each medication was then discussed to take into account the characteristics of remote military operations.
CONCLUSIONS:
In tactical situations, in association with haemostatic procedures (tourniquet, suture, etc), tranexamic acid should be the first medication used according to the current guidelines. The use of fibrinogen concentrate should also be considered for patients in haemorrhagic shock, especially if point-of-care (POC) testing of haemostasis or shock severity is available. If POC evaluation is not available, it seems reasonable to still administer this treatment after clinical assessment, particularly if the evacuation is delayed. In this situation, lyophilised plasma may also be given as a resuscitation fluid while respecting permissive hypotension. Whole blood transfusion in the field deserves special attention.
In addition to the aforementioned treatments, if the field care is prolonged, whole blood transfusion must be considered if it does not delay the evacuation
| Tags : coagulopathie, transfusion
02/01/2017
Coagulopathy/ Eléments de compréhension
Advances in the understanding of trauma-induced coagulopathy
Chang R. et al Blood. 2016;128(8):1043-1049
Ten percent of deaths worldwide are due to trauma, and it is the third most common cause of death in the United States. Despite a profound upregulation in procoagulant mechanisms, one-quarter of trauma patients present with laboratorybased evidence of trauma-induced coagulopathy (TIC), which is associated with poorer outcomes including increased mortality. The most common causes of death after trauma are hemorrhage and traumatic brain injury (TBI). The management of TIC has significant implications in both because many hemorrhagic deaths could be preventable, and TIC is associated with progression of intracranial injury after TBI. This review covers the most recent evidence and advances in our understanding of TIC, including the role of platelet dysfunction, endothelial activation, and fibrinolysis. Trauma induces a plethora of biochemical and physiologic changes, and despite numerous studies reporting differences in coagulation parameters between trauma patients and uninjured controls, it is unclear whether some of these differences may be “normal” after trauma. Comparisons between trauma patients with differing outcomes and use of animal studies have shed some light on this issue, but much of the data continue to be correlative with causative links lacking. In particular, there are little data linking the laboratory-based abnormalities with true clinically evident coagulopathic bleeding. For these reasons, TIC continues to be a significant diagnostic and therapeutic challenge.
| Tags : coagulopathie
15/12/2016
Sang total: Pas que chaud, de banque aussi
Coagulation function of stored whole blood is preserved for 14 days in austere conditions: A ROTEM feasibility study during a Norwegian antipiracy mission and comparison to equal ratio reconstituted blood.
BACKGROUND:
Formulation of a medical preparedness plan for treating severely bleeding casualties during naval deployment is a significant challenge because of territory covered during most missions. The aim of this study was to evaluate the concept of "walking blood bank" as a supportable plan for supplying safe blood and blood products.
METHODS:
In 2013, the Royal Norwegian Navy conducted antipiracy operations from a frigate, beginning in the Gulf of Aden and ending in the Indian Ocean. Crews were on 24-hour emergency alert in preparation for an enemy assault on the frigate. Under an approved command protocol, a "walking blood bank," using crew blood donations, was established for use on board and on missions conducted in rigid-hulled inflatable boats, during which freeze-dried plasma and leukoreduced, group O low anti-A/anti-B titer, cold-stored whole blood were stored in Golden Hour Boxes. Data demonstrating the ability to collect, store, and provide whole blood were collected to establish feasibility of implementing a whole blood-focused remote damage-control resuscitation program aboard a naval vessel. In addition, ROTEM data were collected to demonstrate feasibility of performing this analysis on a large naval vessel and to also measure hemostatic efficacy of cold-stored leukoreduced whole blood (CWB) stored during a period of 14 days. ROTEM data on CWB was compared with reconstituted whole blood.
RESULTS:
Drills simulating massive transfusion activation were conducted, in which 2 U of warm fresh whole blood with platelet sparing leukoreduction were produced in 40 minutes, followed by collection of two additional units at 15-minute increments. The ROTEM machine performed well during ship-rolling, as shown by the overlapping calculated and measured mechanical piston movements measured by the ROTEM device. Error messages were recorded in 4 (1.5%) of 267 tests. CWB yielded reproducible ROTEM results demonstrating preserved fibrinogen function and platelet function for at least 3.5 weeks and 2 weeks, respectively. The frequency of ROTEM tests were as follows: EXTEM (n = 88), INTEM (n = 85), FIBTEM (n = 82), and APTEM (n = 12). CWB results were grouped. Compared with Days 0 to 2, EXTEM maximum clot firmness was significantly reduced, beginning on Days 10 to 14; however, results through that date remained within reference ranges and were comparable with the EXTEM maximum clot firmness for the reconstituted whole blood samples containing Day 5 room temperature-stored platelets.
CONCLUSION:
A "walking blood bank" can provide a balanced transfusion product to support damage-control resuscitation/remote damage-control resuscitation aboard a frigate in the absence of conventional blood bank products. ROTEM analysis is feasible to monitor damage-control resuscitation and blood product quality. ROTEM analysis was possible in challenging operational conditions.
| Tags : coagulopathie, transfusion
14/12/2016
Fibrinogène: Moins de décès
Pre-emptive administration of fibrinogen concentrate contributes to improved prognosis in patients with severe trauma
Yamamoto K, et al. Trauma Surg Acute Care Open 2016;1:1–5
---------------------------------------------------------
Bien que cela reste encore discuté, l'apport précoce de fibrinogène améliorerait la survie des traumatisés sévères. Cette publication est du moins en faveur de cette hypothèse.
---------------------------------------------------------
Background
Patients with severe trauma often present with critical coagulopathy, resulting in impaired hemostasis, massive hemorrhage, and a poor survival prognosis. The efficacy of hemostatic resuscitation in correcting coagulopathy and restoring tissue perfusion has not been studied. We assessed a novel approach of pre-emptive administration of fibrinogen concentrate to improve critical coagulopathy in patients with severe trauma.
Methods
We retrospectively compared blood transfusion volumes and survival prognosis between three groups of patients with trauma, with an Injury Severity Score (ISS) ≥26 over three consecutive periods: group A, no administration of fibrinogen concentrate; group B, administration of 3 g of fibrinogen concentrate after evaluation of trauma severity and a plasma fibrinogen level <1.5 g/L; group C, pre-emptive administration of 3 g of fibrinogen concentrate immediately on patient arrival based on prehospital information, including high-severity injury or assessed
need for massive transfusion before measurement of fibrinogen.
Results
∼56% of patients with an ISS ≥26 and transfused with red blood cell concentrates ≥10 units, had hypofibrinogenemia (fibrinogen <1.5 g/L) on arrival. Patients who received fibrinogen concentrate in group C showed significantly higher fibrinogen levels after treatment with this agent than those in group B (2.41 g/L vs 1.88 g/L; p=0.01). Although no significant difference was observed in blood transfusion volumes between the groups, the 30-day survival of patients in group C (all, and those with an ISS ≥26) was significantly better than in group A ( p<0.05). The 48-hour mortality rate in patients with an ISS ≥26 was significantly lower in group C than in group A (8.6% vs 22.9%; p=0.005). Further, among patients with an ISS ≥41, the overall mortality was significantly lower in group C than in group A (20% vs 50%; p=0.02).
Conclusion
Pre-emptive administration of fibrinogen concentrate for patients with trauma with critical coagulopathy may contribute to improved survival.
| Tags : coagulopathie
27/06/2016
Albumine 20% plutôt que fibrinogène ?
Influences of limited resuscitation with plasma or plasma protein solutions on hemostasis and survival of rabbits with noncompressible hemorrhage
Kheirabadi BS et Al. J Trauma Acute Care Surg. 2016;81: 42–49
--------------------------------
Dans ce travail et alors que l'albumine diluée n'est pas recommandée, les auteurs évoquent la possibilité que les effets favorables d'une réanimation basée sur l'apport de plasma serait lié à l'apport de protéine et en particulier d'albumine qui aurait un effet tampon élevé, réduisant l'acidose métabolique, un des facteurs de la triade létale.
--------------------------------
BACKGROUND:
Plasma infusion with or without red blood cells is the current military standard of care for prehospital resuscitation of combat casualties. We examined possible advantages of early and limited resuscitation with fresh plasma compared with a single plasma protein or crystalloid solutions in an uncontrolled hemorrhage model in rabbits.
METHODS:
Anesthetized spontaneously breathing rabbits (3.3 ± 0.1 kg) were instrumented and subjected to a splenic uncontrolled hemorrhage. Rabbits in shock were resuscitated at 15 minutes with Plasma-Lyte (PAL; 30 mL/kg), PAL + fibrinogen (PAL + F; 30 mL + 100 mg/kg), fresh rabbit plasma (15 mL/kg), or 25% albumin (ALB; 5 mL/kg) solution, all given in two bolus intravenous injections (15 minutes apart) to achieve a mean arterial pressure of 65 mm Hg, n = 8 to 9/group. Animals were monitored for 2 hours or until death, and blood loss was measured. Blood samples and tissues were collected and analyzed.
RESULTS:
There were no differences among groups in baseline measures and their initial bleeding volume at 15 minutes. At 60 minutes after injury, mean arterial pressure was higher with ALB than with crystalloids (PAL or PAL + F), but shock indices were not different despite the large differences in resuscitation volumes. Fibrinogen addition to PAL only increased clot strength. Plasma resuscitation increased survival rate (75%) without significant improvement in coagulation measures. Albumin administration replenished total plasma protein and increased survival rate to 100% (p < .05 vs. crystalloids). No histological adverse events were identified in the vital organs.
CONCLUSIONS:
Fibrinogen administration added to a compatible crystalloid did not improve hemostatic outcomes. Plasma resuscitation increased survival rate; however, its effects did not differ from those obtained with 25% ALB at one-third of the volume. The ALB advantage was consistent with our previous findings in which 5% ALB was used at a volume equal to plasma. The benefit of plasma for resuscitation may be mostly due to its ALB content rather than its coagulation proteins
| Tags : coagulopathie, remplissage
04/06/2016
PLyo: Une révolution ? Pas vraiment, une redécouverte
Pusateri AE et Al. Transfusion. 2016 Apr;56 Suppl 2:S128-39
------------------------------------------
Les nouvelles modalités de transfusion mettent en avant le bénéfice de l'apport précoce de plasma. Les contraintes logistiques liées à l'emploi de plasma frais sont réelles. L'emploi de plasma lyophilisé permet de raccourcir ce délai et peut représenter dans certaines conditions d'isolement la seule source disponibles de fractions coagulantes. Le plasma lyophylisé est un vieux monsieur, mais dont la place est fondamentale. Largement utilisé notamment par l'armée française pendant la guerre d'indocchine, le SSA a maintenu sa production jusqu'à ce que l'épidémie de VIH ne survienne. Depuis les années 1980, le SSA a travaillé sans relâche pour sécuriser un produit qui retrouve la place qui lui est due dans la stratégie transfusionnelle du blessé de guerre (1) Il s'agit donc d'une redécouverte avec un emploi effectif en opération dès 1996 (2), plutôt que de révolution. Le document proposé à la lecture fait le point sur cette historique et les développements à venir. La lecture de ce document ne doit pas faire oublier la réflexion de plus en plus présente sur l'emploi en situation d'isolement de l'intérêt de la transfusion de sang total, seule source de plaquettes, associé au recours à des fractions coagulantes comme le fibrinogène et les complexes prothrombiques. Une telle association représente probablement l'avenir de la réanimation hémostatique préhospitalière (3, 4).
------------------------------------------
Historical dried plasma development Event Selected References
1930s Plasma lyophilization developed in the 1930s.
1940—Large scale production of pooled, lyophilized plasma by both the US and British established for war time use (to meet logistical constraints of whole blood and frozen/liquid plasma).ans les années
1941—Spray dried plasma produced for the Swedish Defense Department. 21 WWII Production 20-22 British produced >500,000 U lyophilized plasma during WWII. US produced >6,000,000 U lyophilized plasma during WWII. US/British distributed world-wide. Sweden produced approximately 17,000 U spray dried plasma for Sweden and Finland.
1945—Hepatitis 23 Hepatitis as a result of plasma transfusion recognized by the end of WWII. Believed that benefits outweighed the risk.
1945-1952—Hepatitis 24 Attempts at pathogen reduction and reducing pool size not successful. Several deaths in clinical studies of ultraviolet irradiated pooled plasma.
1953
—Department of the Army (Circular 73) directed that, because of the risk of serum hepatitis, the higher cost, and the need to use it for the production of specific globulins, plasma would not be used “to support blood volume” unless dextran was not available.
—Serum albumin replaced plasma as primary resuscitative product for US Forces in Korea.
1968—National Research Council Committee on Plasma and Plasma Substitutes recommended that “the use of whole, pooled human plasma be discouraged and even discontinued unless a clear cut case can be made for its unique requirements.”
The French Military Blood Institute produced dried plasma from 1949 to 1984, and provided over 40,000 units to French military forces during the Indochina War. In 1985, production was discontinued due to risk of HIV infection.
| Tags : coagulopathie, transfusion
Acide tranexaminique systématique: La roue tournerait-elle ?
Acute Fibrinolysis Shutdown after Injury Occurs Frequently and Increases Mortality: A Multicenter Evaluation of 2,540 Severely Injured Patients
Moore HB et Al. J Am Coll Surg. 2016 Apr;222(4):347-55.
------------------------------------------------------
La recommandation n°15 de la RFE Portant sur la réanimation du choc hémorragique stipule qu' il est recommandé d'administrer de l'acide tranexamique dès que possible chez les patients traumatisés à la dose de 1 g en bolus intraveineux en 10 min suivi de 1 g perfusé sur 8 h chez les patients traumatisés. Cette administration ne doit pas être initiée au delà de la 3e heure suivant la survenue d'un traumatisme avec choc hémorragique. Si l'étude CRASH-2 a montré que l'acide tranexamique réduisait significativement la mortalité. De nouvelles données disponibles depuis alimentent la discussion (1). Trois états du système de fibrinolyse peuvent être retrouvés: normal, hyperfibrinolyse, inhibition Les deux derniers sont associés à une surmortalité mais la fréquence des états d'inhibition de l'inhibition ne plaiderait pas pour une administration systématique mais ciblée d'exacyl. En qui nous concerne il faut à nouveau insister sur la précocité de l'administration d'exacyl dans l'heure chez les blessés sévères, pour lesquels le bénéfice en terme de mortalité est le plus grand (2). Une réflexion pour un emploi optimisé est en MARCHE (3, 4)
------------------------------------------------------
BACKGROUND:
Fibrinolysis is a physiologic process that maintains microvascular patency by breaking down excessive fibrin clot. Hyperfibrinolysis is associated with a doubling of mortality. Fibrinolysis shutdown, an acute impairment of fibrinolysis, has been recognized as a risk factor for increased mortality. The purpose of this study was to assess the incidence and outcomes of fibrinolysis phenotypes in 2 urban trauma centers.
STUDY DESIGN:
Injured patients included in the analysis were admitted between 2010 and 2013, were 18 years of age or older, and had an Injury Severity Score (ISS) > 15. Admission fibrinolysis phenotypes were determined by the clot lysis at 30 minutes (LY30): shutdown ≤ 0.8%, physiologic 0.9% to 2.9%, and hyperfibrinolysis ≥ 3%. Logistic regression was used to adjust for age, arrival blood pressure, ISS, mechanism, and facility.
RESULTS:
There were 2,540 patients who met inclusion criteria. Median age was 39 years (interquartile range [IQR] 26 to 55 years) and median ISS was 25 (IQR 20 to 33), with a mortality rate of 21%. Fibrinolysis shutdown was the most common phenotype (46%) followed by physiologic (36%) and hyperfibrinolysis (18%). Hyperfibrinolysis was associated with the highest death rate (34%), followed by shutdown (22%), and physiologic (14%, p < 0.001). The risk of mortality remained increased for hyperfibrinolysis (odds ratio [OR] 3.3, 95% CI 2.4 to 4.6, p < 0.0001) and shutdown (OR 1.6, 95% CI 1.3 to 2.1, p = 0.0003) compared with physiologic when adjusting for age, ISS, mechanism, head injury, and blood pressure (area under the receiver operating characteristics curve 0.82, 95% CI 0.80 to 0.84).
CONCLUSIONS:
Fibrinolysis shutdown is the most common phenotype on admission and is associated with increased mortality. These data provide additional evidence of distinct phenotypes of coagulation impairment and that individualized hemostatic therapy may be required.
| Tags : coagulopathie
03/06/2016
The 2015 Remote Damage Control Resuscitation Symposium
| Tags : transfusion, coagulopathie
11/04/2016
PROPPR Study: 1-1-2 aussi bien !
Damage-control resuscitation and emergency laparotomy: Findings from the PROPPR study
Undurraga VJ et AL. J Trauma Acute Care Surg. 2016 Apr;80(4):568-75
BACKGROUND:
The Pragmatic Randomized Optimal Platelet and Plasma Ratios (PROPPR) trial has demonstrated that damage-controlresuscitation, a massive transfusion strategy targeting a balanced delivery of plasma-platelet-red blood cell in a ratio of 1:1:1, results in improved survival at 3 hours and a reduction in deaths caused by exsanguination in the first 24 hours compared with a 1:1:2 ratio. In light of these findings, we hypothesized that patients receiving 1:1:1 ratio would have improved survival after emergency laparotomy.
METHODS:
Severely injured patients predicted to receive a massive transfusion admitted to 12 Level I North American trauma centers were randomized to 1:1:1 versus 1:1:2 as described in the PROPPR trial. From these patients, the subset that underwent an emergency laparotomy, defined previously in the literature as laparotomy within 90 minutes of arrival, were identified. We compared rates and timing of emergency laparotomyas well as postsurgical survival at 24 hours and 30 days.
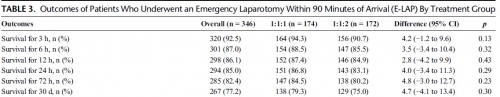
RESULTS:
Of the 680 enrolled patients, 613 underwent a surgical procedure, 397 underwent a laparotomy, and 346 underwent an emergency laparotomy. The percentages of patients undergoing emergency laparotomy were 51.5% (174 of 338) and 50.3% (172 of 342) for 1:1:1 and 1:1:2, respectively (p = 0.20). Median time to laparotomy was 28 minutes in both treatment groups. Among patients undergoing an emergency laparotomy, the proportions of patients surviving to 24 hours and 30 days were similar between treatment arms; 24-hour survival was 86.8% (151 of 174) for 1:1:1 and 83.1% (143 of 172) for 1:1:2 (p = 0.29), and 30-day survival was 79.3% (138 of 174) for 1:1:1 and 75.0% (129 of 172) for 1:1:2 (p = 0.30).
CONCLUSION:
We found no evidence that resuscitation strategy affects whether a patient requires an emergency laparotomy, time to laparotomy, or subsequent survival.
| Tags : coagulopathie, transfusion
28/01/2016
TXA: Interrogations
Les études CRASH2 et MATTERS ont mis en évidence l'intérêt de l'emploi du TXA en traumatologie grave.
Il s'agit d'un dérivé de la lysine qui agit en se liant au plasminogène bloquant ainsi l'interaction plasminogène-fibrine, donc la fibrinolyse du caillot. Le TXA franchit la barrière sang-cerveau, diffuse dans le LCR et le globe oculaire
Persistent malgré tout quelques interrogations en matière d'innocuité persistent. Si l'étude crash2 n' pas montré de risque thromboembolique majeurs, ce n'est pas le cas d' l'étude MATTERS avec environ 10 fois plus d'épisodes thrombo-emboliques en cas d'usage de TXA. Par ailleurs il est rapporté un risque d'hypotension lors de l'administration rapide de TXA et de convulsions lors de l'emploi de posologies élevées. Ceci ne remet pas en cause le recours précoce au TXA dont l'emploi ne doit pas être banalisé et respecter un certain nombre de règles: probabilité forte de coagulopathie traumatique notamment attesté par une hypotension sévère , 1ère dose le plus tôt possible (au mieux dans la première heure) et pas après 3h, deuxième dose dans les 08h00, administration lente pour éviter hypotension, pas de surdosage facteur de crises convulsives, ne pas administrer en même temps/même ligne que du PLYO.
Un certain nombre d'études complémentaires sont en cours:
1. L'étude "Pre-hospital Antifibrinolytics for Traumatic Coagulopathy and Haemorrhage" a pour objectif d'affiner notre connaissance de l'emploi du TXA.
2.L'étude "Design of the Study of Tranexamic Acid during Air Medical Prehospital Transport (STAAMP) Trial: Addressing the Knowledge Gaps" a pour objet d'étuider la mortalité à 30 jours de traumatisés sévères pris en charge par medevac héliportées.
3. L'étude "Tranexamic Acid Mechanisms and Pharmacokinetics In Traumatic Injury (TAMPITI Trial)" vise quand à elle à confirmer un certain nombre d'hypothèses sur le mécanisme d'action.
Par ailleurs, le TXA n'est pas le seul antifibrinolyique utilisable.
| Tags : coagulopathie
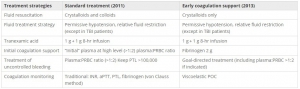
20/12/2015
Coagulopathie: Du fibrinogène avant tout
Trauma-induced coagulopathy: impact of the early coagulation support protocol on blood product consumption, mortality and costs
Nardi G et al. Critical Care (2015) 19:83
------------------------------------
Le damage control resuscitation fait largement appel à l'apport de plasma, concentrés de globules rouges et de plaquettes dans un rapport de 1/1/1 (1). Malgré la relative correction de la coagulopathie la survie à long terme ne semble pas être améliorée (2). Aussi certains proposent d'avoir plutôt recours à l'administration précoce de fibrinogène plutôt que l'administration de plasma. Le travail suivant qui propose l'apport précoce de 2g de fibrinogène est en faveur d'une telle démarche.
------------------------------------
INTRODUCTION:
Hemorrhage is the principal cause of death in the first few hours following severe injury. Coagulopathy is a frequent complication of critical bleeding. A network of Italian trauma centers recently developed a protocol to prevent and treat trauma-induced coagulopathy. A pre-post cohort multicenter study was conducted to assess the impact of the early coagulation support (ECS) protocol on blood products consumption,mortality and treatment costs.
METHODS:
We prospectively collected data from all severely injured patients (Injury Severity Score (ISS) >15) admitted to two trauma centers in 2013 and compared these findings with the data for 2011. Patients transfused with at least 3 units of packed red blood cells (PRBCs) within 24 hours of an accident were included in the study. In 2011, patients with significant hemorrhaging were treated with early administration of plasma with the aim of achieving a high (≥1:2) plasma-to-PRBC ratio. In 2013, the ECS protocol was the treatment strategy. Outcome data, blood product consumption and treatment costs were compared between the two periods.
RESULTS:
The two groups were well matched for demographics, injury severity (ISS: 32.9 in 2011 versus 33.6 in 2013) and clinical and laboratory data on admission. In 2013, a 40% overall reduction in PRBCs was observed, together with a 65% reduction in plasma and a 52% reduction in platelets. Patients in the ECS group received fewer blood products: 6.51 units of PRBCs versus 8.14 units. Plasma transfusions decreased from 8.98 units to 4.21 units (P <0.05), and platelets fell from 4.14 units to 2.53 units (P <0.05). Mortality in 2013 was 13.5% versus 20% in 2011 (13 versus 26 hospital deaths, respectively) (nonsignificant). When costs for blood components, factors and point-of-care tests were compared, a €76,340 saving in 2013 versus 2011 (23%) was recorded.
CONCLUSIONS:
The introduction of the ECS protocol in two Italian trauma centers was associated with a marked reduction in blood product consumption, reaching statistical significance for plasma and platelets, and with a non-significant trend toward a reduction in early and 28-daymortality. The overall costs for transfusion and coagulation support (including point-of-care tests) decreased by 23% between 2011 and 2013.
| Tags : coagulopathie
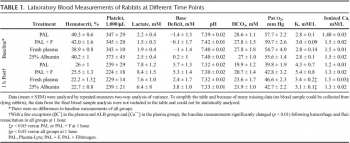
12/11/2015
Et l'albumine ?
Is limited prehospital resuscitation with plasma more beneficial than using a synthetic colloid? An experimental study in rabbits with parenchymal bleeding
Kheirabadi BS et Al. J Trauma Acute Care Surg. 2015;78: 752-759
--------------------------------------
Il existe de grands débats sur la manière optimale d'assurer le remplissage vasculaire des blessés de guerre. Pour certains le fluide de référence est un hydroxyéthylamidon, pour d'autres un cristalloïde isotonique et en ce qui nous concerne les deux avec la mise en avant du sérum salé hypertonique premier suivi d'HEA. Actuellement il existe une tendance à promouvoir une autre stratégie faisant appel pour les blessés les plus graves au plasma voire la transfusion de sang frais. L'étude proposée avait pour objectif de confirmer l'intérêt d'une démarche "plasma premier". Une des surprises a été de constater que ce n'est pas cette dernière qui permettait d'obtenir le meilleur taux de survie mais l'emploi d'albumine, et ce de loin. Ces données expérimentales certes très partielles permettent aux auteurs (?) de rediscuter les conclusions de travaux anciens notamment de l'étude SAFE (1,2). Les solutés d'albumine utilisés par cette dernière ont une osmolarité de 260 mosm/kg (versus 305 mosm/kg pour le sérum salé). Les effets délétères notamment chez le traumatisé crânien pourraient être dus non pas à l'extravasation d'albumine dans le parenchyme cérébral lésé mais à l'hypoosmolarité de l'albumex 4%, ces deux mécanismes concourrant à la plus grande fréquence d'HTIC dans le groupe albumine (3). A méditer
--------------------------------------
BACKGROUND:
Reports of survival benefits of early transfusion of plasma with red blood cells (1:1 ratio) in trauma patients suggest that plasma may be a better fluid to replace Hextend for battlefield resuscitation. We studied possible advantages of prehospital resuscitation with plasma compared with Hextend or albumin in a model of uncontrolled hemorrhage.
METHODS:
Male New Zealand white rabbits (3.3 T 0.1 kg) were anesthetized, instrumented, and subjected to a splenic injury with uncontrolled bleeding. Ten minutes after injury (mean arterial pressure [MAP] G 40 mm Hg), the rabbits received small and equal volumes (15 mL/kg) of rabbit plasma (n = 10), Hextend (n = 10), or 5% human albumin (n = 9) or no fluid. Fluids were administered in two bolus injections (20 minutes apart) and targeted to aMAP of 65 mm Hg. Animals were monitored for 2.5 hours or until death, and their blood losses were measured. Arterial blood samples were collected at different times and analyzed for ABG, CBC, and coagulation tests.
RESULTS:
There were no differences in baseline measures among groups. Splenic injury caused similar hemorrhages (9.1 T 0.4 mL/kg at 10 minutes) and decreased MAP in all subjects. Subsequent resuscitation initiated additional bleeding. At 60 minutes after injury (20 minutes after resuscitation), longer activated partial thromboplastin time and lower fibrinogen concentrations were apparent compared with baseline values with differences among groups. Thrombelastography analysis indicated faster and stronger clot formation with plasma and albumin resuscitation than with Hextend use. Shock indices were increased in all groups, but smaller changes were measured in the albumin group. Total blood loss did not differ among resuscitated rabbits but was higher (p G 0.05) than among nonresuscitated animals. Survival rates were 11% (untreated), 40% (Hextend and plasma), and 89% (albumin, p G 0.05).
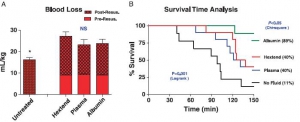
CONCLUSION:
Resuscitation with plasma or albumin better preserved coagulation function than did Hextend. However, despite these improvements, plasma resuscitation did not reduce blood loss or improve survival, while albumin administration seemed beneficial
| Tags : remplissage, coagulopathie
10/11/2015
Dépakine chez le blessé cranien en choc ?
Treatment with a histone deacetylase inhibitor, valproic acid, is associated with increased platelet activation in alarge animal model of traumatic brain injury and hemorrhagic shock
Dekker SE et Al. J Surg Res. 2014 Jul;190(1):312-8
---------------------------
Le concept du damaged control resuscitation fait appel en partie à de nouvelles modalités transfusionnelles et d'emploi de fractions coagulantes. D'autres approches sont possibles comme celles visant à restuarer la fonction plaquettaire. C'est ce que permettrait l'adminsitration de médicaments appartenant à la classe des inhibiteurs des histone deacetylase et dont les effets neuroprotecteurs pourraient ainsi être mis à profit. Le document proposé semble conforter cette approche.
---------------------------
BACKGROUND:
We have previously shown that resuscitation with fresh frozen plasma (FFP) in a large animal model of traumatic brain injury (TBI) and hemorrhagic shock (HS) decreases the size of the brain lesion, and that addition of a histone deacetylase inhibitor, valproic acid (VPA), provides synergistic benefits. In this study, we hypothesized that VPA administration would be associated with a conservation of platelet function as measured by increased platelet activation after resuscitation.
MATERIALS AND METHODS:
Ten swine (42-50 kg) were subjected to TBI and HS (40% blood loss). Animals were left in shock for 2 h before resuscitation with either FFP or FFP+VPA (300 mg/kg). Serum levels of platelet activation markers transforming growth factor beta, CD40 L, P-selectin, and platelet endothelial cell adhesion molecule (PECAM) 1 were measured at baseline, postresuscitation, and after a 6-h observation period. Platelet activation markers were also measured in the brain whole cell lysates and immunohistochemistry.
RESULTS:
Circulating P-selectin levels were significantly higher in the FFP+VPA group compared with the FFP alone group (70.85±4.70 versus 48.44±7.28 ng/mL; P<0.01). Likewise, immunohistochemistry data showed elevated P-selectin in the VPA treatment group (22.30±10.39% versus 8.125±3.94%, P<0.01). Serum sCD40L levels were also higher in the FFP+VPA group (3.21±0.124 versus 2.38±0.124 ng/mL; P<0.01), as was brainsCD40L levels (1.41±0.15 versus 1.22±0.12 ng/mL; P=0.05). Circulating transforming growth factor beta levels were elevated in the FFP+VPA group, but this did not reach statistical significance (11.20±1.46 versus 8.09±1.41 ng/mL; P=0.17). Brain platelet endothelial cell adhesion molecule 1 levels were significantly lower in the FFP+VPA group compared with the FFP group (5.22±2.00 pg/mL versus 7.99±1.13 pg/mL; P=0.03).
CONCLUSIONS:
In this clinically relevant large animal model of combined TBI+HS, the addition of VPA to FFP resuscitation results in an early upregulation of platelet activation in the circulation and the brain. The previously observed neuroprotective effects of VPA may be due to a conservation of platelet function as measured by a higher platelet activation response after resuscitation.
| Tags : tbi, coagulopathie
09/11/2015
Plaie cérébrale et coagulopathie
Quelques faits
1. Elle est fréquente voire très fréquente: Greuters et al. Critical Care 2011 15:R2 doi:10.1186/cc9399

2. Elle est + fréquente en cas d'hypoTA: Wafaisade Neurocrit Care. 2010 Apr;12(2):211-9
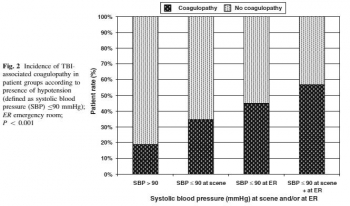
3. Elle est de mauvais pronostic: J Emerg Trauma Shock. 2013 Jul-Sep; 6(3): 180–185

4. Elle est mise en évidence plutôt par thromboélastographie (r TEG) : Sixta al., J Neurol Neurophysiol 2014, 6:5
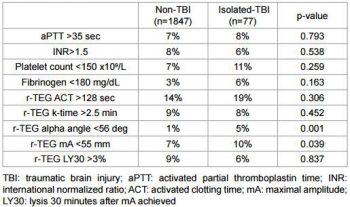
| Tags : coagulopathie