26/02/2026
Pst Hémostatique et temérpatures extrêmes: C'est OK
12/12/2025
TXA: 2g IV si trauma crânien
Optimal dose of tranexamic acid in traumatic brain injury: Systematic review and network meta-analysis of randomized controlled trials
Shu U et Al Journal of Trauma and Acute Care Surgery 98(5):p 816-823, May 2025.
BACKGROUND
Tranexamic acid (TXA) has been used to treat traumatic brain injury (TBI); however, no definitive conclusions have been drawn regarding its effectiveness or dosage. This study evaluated the optimal TXA dose for treating TBI using a network meta-analysis (NMA).
METHODS
Five databases were searched for peer-reviewed randomized controlled trials (RCTs) published from inception to May 2024. The inclusion criteria were as follows: (1) RCTs, (2) patients older than 1 month with TBI, (3) interventions of TXA and control, (4) primary outcomes of mortality and poor neurological outcomes and secondary outcomes of vascular occlusive events, and (5) full-text peer-reviewed articles. Two reviewers independently screened and extracted the data and assessed the risk of bias. Frequency-based NMA was performed using the Grading of Recommendations, Assessment, Development, and Evaluation working-group approach.
RESULTS
We included 10 RCTs comprising 11,237 patients with TBI. Placebo showed higher mortality compared with that of a 2-g bolus of TXA (risk ratio, 1.53; 95% confidence interval, 1.08–2.17). Higher mortality was observed with a 1-g bolus of TXA followed by 1-g maintenance TXA compared with that of a 2-g bolus of TXA (risk ratio, 1.44; 95% confidence interval, 1.02–2.03). No significant differences in poor neurological outcomes or vascular occlusive events were observed between the treatment groups.

CONCLUSION
Placebo and a 1-g bolus followed by 1-g maintenance TXA were associated with higher mortality rates than those of a 2-g bolus of TXA. No difference in vascular occlusive events was observed with either treatment, indicating that our NMA recommends 2 g of TXA. However, the data for the 2-g bolus of TXA were from a single study, and further research is needed to draw definitive conclusions.
Récapitulatif sur le remplissage vasculaire/Transfusion
Fluid resuscitation in trauma: What you need to know
Navpreet D et Al. Journal of Trauma and Acute Care Surgery 98(1):p 20-29, January 2025.
There have been numerous changes in resuscitation strategies for severely injured patients over the last several decades. Certain strategies, such as aggressive crystalloid resuscitation, have largely been abandoned because of the high incidence of complications and worsening of trauma-induced coagulopathy. Significant emphasis has been placed on restoring a normal coagulation profile with plasma or whole blood transfusion. In addition, the importance of the lethal consequences of trauma-induced coagulopathy, such as hyperfibrinolysis, has been easily recognized by the use of viscoelastic testing, and its treatment with tranexamic acid has been extensively studied. Furthermore, the critical role of early intravenous calcium administration, even before blood transfusion administration, has been emphasized.

Other adjuncts, such as fibrinogen supplementation with fibrinogen concentrate or cryoprecipitate and prothrombin complex concentrate, are being studied and incorporated in some of the institutional massive transfusion protocols. Finally, balanced blood component transfusion (1:1:1 or 1:1:2) and whole blood have become commonplace in trauma centers in North America. This review provides a description of recent developments in resuscitation and a discussion of recent innovations and areas for future investigation.
07/12/2025
Vers un nouveau Plyo
Efficacy and safety of novel freeze-dried plasma products in a porcine combat casualty model
Dufour-Gaume F et Al. Transfusion. 2024 Sep;64(9):1670-1682
-----------
Le Plyo est un des produits phares développé par le CTSA. Les recherches continuent pour améliorer les caractérisques. C'est le sens de ce tavail qui porte sur du Plyo plus concentré ou enrichi en lyophylisat de plaquettes. Ce dernier produit parait particulièrement intéressant. l'industrie pharmaceutique n'est pas en reste puisqu'elle propose maintenant des alternatives au PLyo standard comme l'OctaplastLG considéré comme un médicament et non plus un dérivé du sang.
-----------
Background
Hemorrhagic shock is well documented as a leading cause of preventable fatalities among military casualties. During military operations plasma can be transfused while waiting for whole blood. This study was conducted to assess the safety and efficacy of two new freeze-dried plasma formulations in a porcine model of traumatic hemorrhagic shock.
Study Design and Methods
In the face of species-specific transfusion, transfusible blood products were derived from porcine sources. The efficacy of three lyophilized plasma (LP) formulations was evaluated: lyophilized plasma (LP), concentrated lyophilized plasma (CLP), and platelet-rich concentrated lyophilized plasma (PCLP). Pigs were subjected to multi-trauma and hemorrhagic shock. Ninety minutes post-shock induction, the animals were treated with one of the three lyophilized products. Monitoring included systolic blood pressure and cardiac output. Point-of-care and laboratory diagnostic tests were used to assess renal function, real-time hemostasis (ROTEM), and coagulation. Histological examinations of kidney, lung, and muscle tissues were conducted 4 h after shock induction.
Results
CLP and PCLP significantly improved systolic blood pressure and cardiac output and positively influenced base excess, creatinine, various ROTEM, and coagulation markers compared with standard LP without histologic modification. No adverse effect was associated with the transfusion of any of the plasma products throughout the experimental procedures.
Conclusion
Both CLP and PCLP exhibit promising therapeutic potential for managing hemorrhagic shock in scenario where whole blood supplies are limited. However, the distinct physiological and coagulation characteristics of the swine model necessitate further investigation using humanized preclinical models to fully understand their clinical applicability and constraints
Plasma: Un point en 2024
Dried Plasma for Major Trauma: Past, Present, and Future
03/06/2025
Drone et transfusion à l'avant
Massive transfusion on the combat field using autonomous drones: A case report
Türkoğlu B et Al . Transfusion. 2025 May 10. doi: 10.1111/trf.18279.
----------------------------------
Le transport de dérivés sanguins par drone est effectif et évalué depuis plusieurs années (1) notamment au Rwanda (2). Le cas rapporté est celui d'un blessé par IED. Le transport vers un role 1 est certainement d'un grand intérêt (4), cependant l'utilisation à grande échelle lors de conflit à haute intensité est certainement un sujet d'intérêt mais très probablement avec des objectifs plus larges et différents (5, 6)
Pour aller plus loin sur l'emploi des drones en médecine préhospitalière (7)
----------------------------------
Background:
Hemorrhage is a leading cause of preventable deaths in combat settings, requiring rapid blood transfusion to improve survival. While the feasibility of drone-assisted medical logistics has been explored, its practical application in battlefield transfusion remains unreported.
Study design and methods:
This case report describes the first documented massive transfusion in a combat environment using an autonomous unmanned aerial vehicle (UAV). A 27-year-old soldier sustained severe lower limb injuries from an improvised explosive device explosion and developed hemorrhagic shock in a remote battlefield location where adverse weather conditions prevented immediate evacuation. In response, 6 units of whole blood and 2 units of fresh frozen plasma were transported via UAV, enabling prehospital transfusion under telemedicine supervision.
Results:
The UAV successfully delivered blood products on two consecutive flights, ensuring early resuscitation and stabilization despite delayed evacuation. The casualty's vital signs improved post-transfusion, and surgical interventions were successfully performed following hospital admission. This case demonstrates the feasibility of drone-assisted blood product transport in prolonged field care scenarios.
Discussion:
To our knowledge, this is the first reported case of a combat casualty receiving a UAV-facilitated massive transfusion in an operational setting. While UAV-based medical logistics offers a rapid and reliable alternative for remote trauma care, challenges remain in regulatory implementation, blood product stability, and integration into standardized protocols. Further controlled studies are needed to optimize UAV-assisted transfusion strategies and their potential expansion into civilian and disaster response settings.
| Tags : drone
12/11/2024
PLyo: Oui mais sans hémodilution par les crstalloïdes
Prehospital Lyophilized Plasma Transfusion for Trauma-Induced Coagulopathy in Patients at Risk for Hemorrhagic Shock. A Randomized Clinical Trial
Jost D. et Al.JAMA Netw Open. 2022;5(7):e2223619.
-----------------
Le recours au plasma fait partie de l'arsenal thérapeutique de la prise en charge des traumatisés sévères en choc hémorragique. Comme l'étude REPHILL (1), Ce travail ne met pas en évidence d'amélioration de la survie à 28 jours. Les délais de transport et le degré d'hémodilution lié au recours aux solutés salés expliquent peut être cela (2)
Key Points
Question Does prehospital transfusion of lyophilized plasma result in a lower incidence of trauma-induced coagulopathy at hospital admission compared with standard care with normal saline infusion in patients at risk for hemorrhagic shock after trauma?
Findings This multicenter randomized clinical trial included 150 patients with trauma who were treated in a prehospital setting. Median international normalized ratio at hospital admission, massive transfusion rate, 30-day survival, and adverse events did not significantly differ between lyophilized plasma transfusion and standard care.
Meaning These findings suggest that prehospital lyophilized plasma transfusion is a feasible and safe procedure for patients who are at risk for hemorrhagic shock, although there is a lack of evidence regarding its ability to prevent trauma-induced coagulopathy.
Abstract
Importance Blood transfusion is a mainstay of therapy for trauma-induced coagulopathy, but the optimal modalities for plasma transfusion in the prehospital setting remain to be defined.
Objective To determine whether lyophilized plasma transfusion can reduce the incidence of trauma-induced coagulopathy compared with standard care consisting of normal saline infusion.
Design, Setting, and Participants This randomized clinical trial was performed at multiple centers in France involving prehospital medical teams. Participants included 150 adults with trauma who were at risk for hemorrhagic shock and associated coagulopathy between April 1, 2016, and September 30, 2019, with a 28-day follow-up. Data were analyzed from November 1, 2019, to July 1, 2020.
Intervention Patients were randomized in a 1:1 ratio to receive either plasma or standard care with normal saline infusion (control).
Main Outcomes and Measures The primary outcome was the international normalized ratio (INR) on arrival at the hospital. Secondary outcomes included the need for massive transfusion and 30-day survival. As a safety outcome, prespecified adverse events included thrombosis, transfusion-related acute lung injury, and transfusion-associated circulatory overload.
Results Among 150 randomized patients, 134 were included in the analysis (median age, 34 [IQR, 26-49] years; 110 men [82.1%]), with 68 in the plasma group and 66 in the control group. Median INR values were 1.21 (IQR, 1.12-1.49) in the plasma group and 1.20 (IQR, 1.10-1.39) in the control group (median difference, −0.01 [IQR, −0.09 to 0.08]; P = .88). The groups did not differ significantly in the need for massive transfusion (7 [10.3%] vs 4 [6.1%]; relative risk, 1.78 [95% CI, 0.42-8.68]; P = .37) or 30-day survival (hazard ratio for death, 1.07 [95% CI, 0.44-2.61]; P = .89). In the full intention-to-treat population (n = 150), the groups did not differ in the rates of any of the prespecified adverse events.
Conclusions and Relevance In this randomized clinical trial including severely injured patients at risk for hemorrhagic shock and associated coagulopathy, prehospital transfusion of lyophilized plasma was not associated with significant differences in INR values vs standard care with normal saline infusion. Nevertheless, these findings show that lyophilized plasma transfusion is a feasible and safe procedure for this patient population.
24/09/2024
Plasma; Et l'OctaplasLG ?
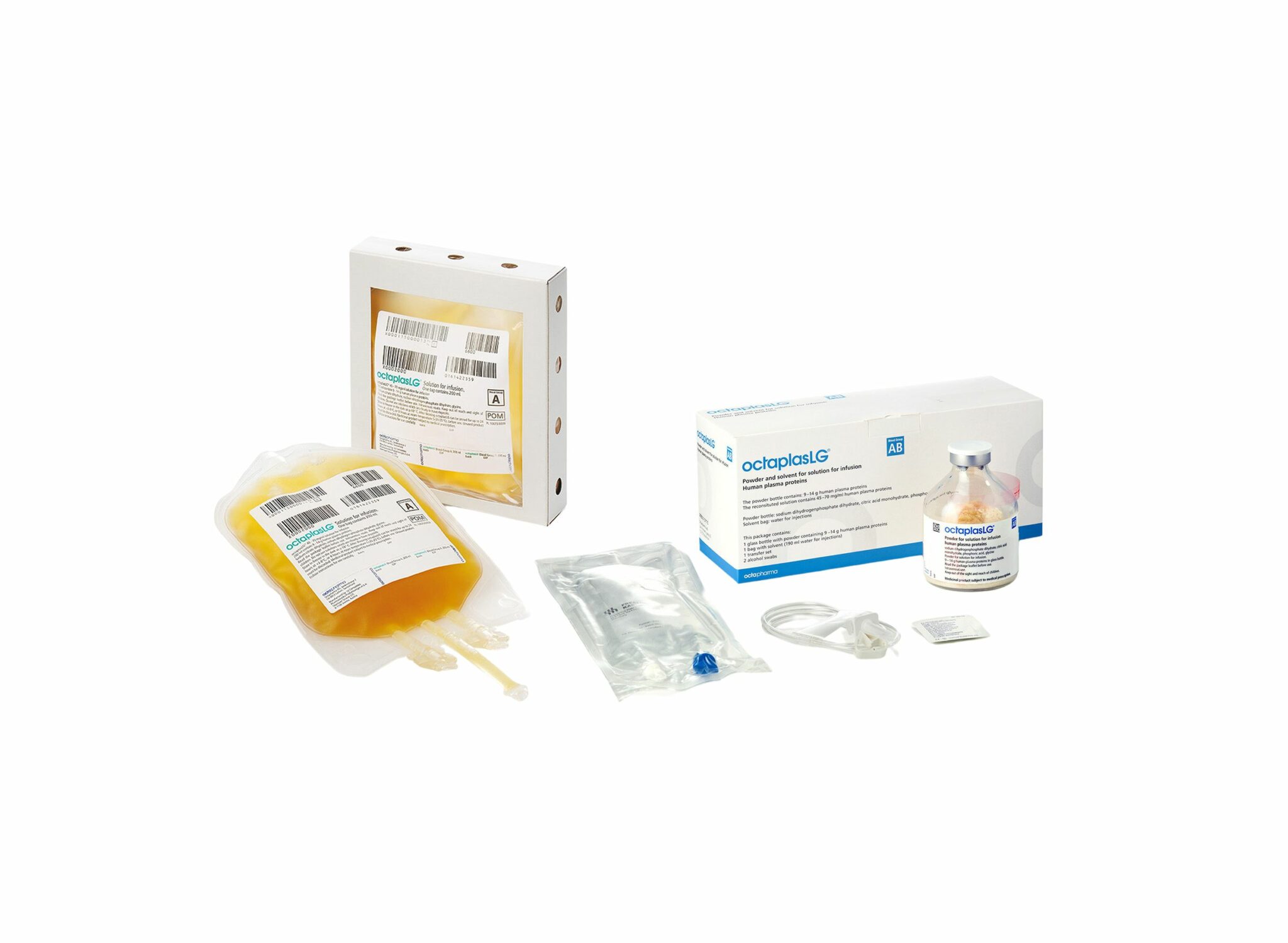
Une alternative HOSPITALIERE au PLyo du fait d'une production actuelle limitée ? VIent d'obtenir son agréement en Europe. Plus simple en gestion car considéré comme un médicament et non un produit sanguin. Il est fourni en présentations distinctes, selon les groupes sanguins. L’administration d’OCTAPLASLG doit respecter les règles de compatibilité des groupes sanguins ABO. En cas d’urgence, OCTAPLASLG du groupe AB peut être considéré comme plasma universel dans la mesure où il peut être administré à tous les patients, indépendamment de leur groupe sanguin. C'est donc dans cette forme qu'il peut être utilisé en préhospitalier comme le Plyo.
Sa place reste à préciser
10/06/2024
Fibrinogène précoce: Moins de transfusion ?
Hemostatic effect of fibrinogen concentrate on traumatic massive hemorrhage: a propensity score matching study
Heo HJ et al. Trauma Surg Acute Care Open. 2024 Jan 30;9(1):e001271.
Background:
Fibrinogen concentrate (FC) can be administered during massive transfusions to manage trauma-induced coagulopathy. However, its effectiveness in survival remains inconclusive due to scarce high-level evidence. This study aimed to investigate the hemostatic effects of FC regarding mortality in massive hemorrhage caused by trauma.
Methods:
This retrospective study analyzed 839 patients who received massive transfusions (red blood cells (RBCs) ≥5 units in 4 hours or ≥10 units in 24 hours) at a level I trauma center between 2015 and 2022. Patients who were transferred to other hospitals or were deceased upon arrival, suffered or died from severe brain injury, and were aged 15 years or less were excluded (n=334). 1:2 propensity score matching was performed to compare the 'FC (+)' group who had received FC in 24 hours (n=68) with those who had not ('FC (-)', n=437). The primary outcome was mortality, and the secondary outcomes included transfusion volume.
Results:
The variables for matching included vital signs, injury characteristics, prehospital time, implementation of resuscitative endovascular balloon occlusion of the aorta, and blood gas analysis results. The administration of FC did not significantly reduce or predict mortality (in-hospital, 24 hours, 48 hours, or 7 days). The FC (-) group received more units of RBC (25.69 units vs. 16.71 units, p<0.001, standardized mean difference [SMD] 0.595), fresh frozen plasma (16.79 units vs. 12.91 units, p=0.023, SMD 0.321), and platelets (8.76 units vs. 5.46 units, p=0.002, SMD 0.446) than the FC (+) group.
Conclusion:
The use of FC did not show survival benefits but reduced transfusion requirements in traumatic massive hemorrhages, highlighting a need for future investigations. In the future, individualized goal-directed transfusion with FC may play a significant role in treating massive bleeding.
PLyo après reconstitution ?
Post-Reconstitution Hemostatic Stability Profiles of Canadian and German Freeze-Dried Plasma
Henry T Peng H et Al. Life (Basel). 2024 Jan 24;14(2):172. doi: 10.3390/life14020172.
Despite the importance of the hemostatic properties of reconstituted freeze-dried plasma (FDP) for trauma resuscitation, few studies have been conducted to determine its post-reconstitution hemostatic stability. This study aimed to assess the short- (≤24 h) and long-term (≥168 h) hemostatic stabilities of Canadian and German freeze-dried plasma (CFDP and LyoPlas) after reconstitution and storage under different conditions. Post-reconstitution hemostatic profiles were determined using rotational thromboelastometry (ROTEM) and a Stago analyzer, as both are widely used as standard methods for assessing the quality of plasma. When compared to the initial reconstituted CFDP, there were no changes in ROTEM measurements for INTEM maximum clot firmness (MCF), EXTEM clotting time (CT) and MCF, and Stago measurements for prothrombin time (PT), partial thromboplastin time (PTT), D-dimer concentration, plasminogen, and protein C activities after storage at 4 °C for 24 h and room temperature (RT) (22-25 °C) for 4 h. However, an increase in INTEM CT and decreases in fibrinogen concentration, factors V and VIII, and protein S activities were observed after storage at 4 °C for 24 h, while an increase in factor V and decreases in antithrombin and protein S activities were seen after storage at RT for 4 h. Evaluation of the long-term stability of reconstituted LyoPlas showed decreased stability in both global and specific hemostatic profiles with increasing storage temperatures, particularly at 35 °C, where progressive changes in CT and MCF, PT, PTT, fibrinogen concentration, factor V, antithrombin, protein C, and protein S activities were seen even after storage for 4 h.
We confirmed the short-term stability of CFDP in global hemostatic properties after reconstitution and storage at RT, consistent with the shelf life of reconstituted LyoPlas. The long-term stability analyses suggest that the post-reconstitution hemostatic stability of FDP products would decrease over time with increasing storage temperature, with a significant loss of hemostatic functions at 35 °C compared to 22 °C or below. Therefore, the shelf life of reconstituted FDP should be recommended according to the storage temperature.
03/04/2023
Sang de banque sur pied: Mieux si on est entraîné
A prospective assessment of the medic autologous blood transfusion skills for field transfusion preparation
Steven G Schauer SG et Al. Transfusion. 2023 Mar 27. doi: 10.1111/trf.17325.
Background: Data demonstrate benefit from blood product administration near point-of-injury (POI). Fresh whole blood transfusion from a pre-screened donor provides a source of blood at the POI when resources are constrained. We captured transfusion skills data for medics performing autologous blood transfusion training.
Methods: We conducted a prospective, observational study of medics with varying levels of experience. Inexperienced medics were those with minimal or no reported experience learning the autologous transfusion procedures, versus reported experience among special operations medics. When available, medics were debriefed after the procedure for qualitative feedback. We followed them up to 7 days for adverse events.
Results: The median number of attempts for inexperienced and experienced medics was 1 versus 1 (interquartile range 1-1 for both, p=0.260). The inexperienced medics had a slower median time to needle venipuncture access for donation of 7.3 versus 1.5 minutes, needle removal after clamping time of 0.3 versus 0.2 minutes, time to bag preparation of 1.9 versus 1.0 minutes, time to IV access for reinfusion of 6.0 versus 3.0 minutes, time to transfusion completion of 17.3 versus 11.0 minutes, and time to IV removal of 0.9 versus 0.3 minutes (all p<0.05). We noted one administrative safety event in which allogeneic transfusion occurred. No major adverse events occurred. Qualitative data saturated around the need for quarterly training.
Conclusions: Inexperienced medics have longer procedure times when training autologous whole blood transfusion skills. This data will help establish training measures of performance for skills optimization when learning this procedure.
01/12/2022
Durée de conservation du sang en milieu hostile
Whole Blood Storage Temperature Investigation in Austere Environments
Cesar OA et Al. J Spec Oper Med. 2022 Sep 19;22(3):19-21.
Introduction:
Military medical research has affirmed that early administration of blood products and timely treatment save lives. The US Navy's Expeditionary Resuscitative Surgical System (ERSS) is a Role 2 Light Maneuver team that functions close to the point of injury, administering blood products and providing damage-control resuscitation and surgery. However, information is lacking on the logistical constraints regarding provisions for and the stability of blood products in austere environments.
Methods:
ERSS conducted a study on the United States Central Command (USCENTCOM) area of responsibility. Expired but properly stored units of stored whole blood (SWB) were subjected to five different storage conditions, including combinations of passive and active refrigeration. The SWB was monitored continuously, including for external ambient temperatures. The time for the SWB to rise above the threshold temperature was recorded.
Results:
The main outcome of the study was the time for the SWB to rise above the recommended storage temperature. Average ambient temperature during the experiment involving conditions 1 through 4 was 25.6°C (78.08°F). Average ambient temperature during the experiment involving condition 5 was 34.8°C (94.64°F). Blood temperature reached the 6°C (42.8°F) threshold within 90 minutes in conditions 1 and 2, which included control and chemically activated ice packs in the thermal insulated chamber (TIC). Condition 2 included prechilling the TIC in a standard refrigerator to 4°C (39.2°F), which kept the units of SWB below the threshold temperature for 490 minutes (approximately 8 hours). Condition 4 entailed prechilling the TIC in a standard freezer to 0.4°C (32.72°F), thus keeping the units of SWB below threshold for 2,160 minutes (i.e., 36 hours). Condition 5 consisted of prechilling the TIC to 3.9°C (39.02°F) in the combat blood refrigerator, which kept the SWB units below the threshold for 780 minutes (i.e., 13 hours), despite a higher average ambient temperature of almost +10°C (50°F).
Conclusion:
Combining active and passive refrigeration methods will increase the time before SWB rises above the threshold temperature. We demonstrate an adaptable approach of preserving blood product temperature despite refrigeration power failure in austere settings, thereby maintaining mission readiness to increase the survival of potential casualties.
France: Transfusion en Opex
An observational study of the blood use in combat casualties of the French Armed Forces, 2013–2021
Py N. et Al. Transfusion. 2022;1–14.
Background:
The French Armed Forces conduct asymmetric warfare in the Sahara-Sahel Strip. Casualties are treated with damage control resuscitation to the extent possible. Questions remain about the feasibility and sustainability of using blood for wider use in austere environments.
Methods:
We performed a retrospective analysis of all French military trauma patients transfused after injury in overseas military operations in Sahel-Saharan Strip, from the point of injury, until day 7, between January 11, 2013 to December 31, 2021.
Results:
Forty-five patients were transfused. Twenty-three (51%) of them required four red blood cells units (RBC) or more in the first 24H defining a severe hemorrhage. The median blood product consumption within the first 48 h, was 8 (IQR [3; 18]) units of blood products (BP) for all study population but up to 17 units (IQR [10; 27.5]) for the trauma patients with severe hemor- rhage. Transfusion started at prehospital stage for 20 patients (45%) and included several blood products: French lyophilized plasma, RBCs, and whole blood. Patients with severe hemorrhage required a median of 2 [IQR 0; 34] fur- ther units of BP from day 3 to day 7 after injury. Eight patients died in theater, 4 with severe hemorrhage and these 4 used an average of 12 products at Role 1 and 2.
Conclusion:
The transfusion needs were predominant in the first 48 h after the injury but also continued throughout the first week for the most severe trauma patients. Importantly, our study involved a low-intensity conflict, with a small number of injured combatants.
12/11/2022
Sang complet: OUI CHAUD et dans les 6 1ères heures.
Whole blood at the tip of the spear: A retrospective cohort analysis of warm fresh whole blood resuscitation vert: Dans les 06sus component therapy in severely injured combat casualties
Surgery . 2022 Feb;171(2):518-525. Gurney JM et Al.
Background
Methods
Results
Conclusion
21/10/2022
Plyo intraosseux ? Pas si simple
Intraosseous administration of freeze-dried plasma in the prehospital setting
Rittblat M et Al. Isr Med Assoc J. 2022 Sep;24(9):591-595.
Background:
Freeze dried plasma (FDP) is a commonly used replacement fluid in the prehospital setting when blood products are unavailable. It is normally administered via a peripheral intravenous (PIV) line. However, in severe casualties, when establishing a PIV is difficult, administration via intraosseous vascular access is a practical alternative, particularly under field conditions.
Objectives:
To evaluate the indications and success rate of intraosseous administration of FDP in casualties treated by the Israel Defense Forces (IDF).
Methods:
A retrospective analysis of data from the IDF-Trauma Registry was conducted. It included all casualties treated with FDP via intraosseous from 2013 to 2019 with additional data on the technical aspects of deployment collected from the caregivers of each case.
Results:
Of 7223 casualties treated during the study period, intravascular access was attempted in 1744; intraosseous in 87 of those. FDP via intraosseous was attempted in 15 (0.86% of all casualties requiring intravascular access). The complication rate was 73% (11/15 of casualties).
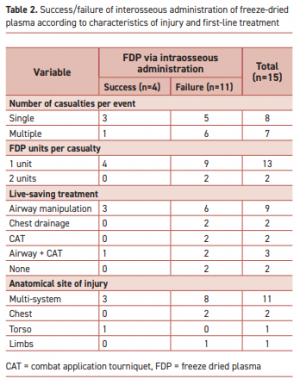
"According to the manufacturer’s specifications, the reconstituted lyophilized plasma product is delivered from a glass bottle with the rate of infusion determined primarily by gravity. This procedure precludes the care providers from applying external pressure to increase the plasma infusion rate. Therefore, the pressure gradient is determined primarily by gravity with respect to height of the bottle."
Complications were more frequent when the event included multiple casualties or when the injury included multiple organs. Of the 11 failed attempts, 5 were reported as due to slow flow of the FDP through the intraosseous apparatus. Complications in the remaining six were associated with deployment of the intraosseous device.
Conclusions:
Administration of FDP via intraosseous access in the field requires a high skill level.
| Tags : intraosseux
04/03/2022
Transfusion de sang frais
Use of Walking Blood Bank at Point of Injury During Combat Operations. A Case Report
Gaddy M et Al. J Spec Oper Med. Winter 2021;21(4):94-98
The US Military Tactical Combat Casualty Care guidelines recommend blood products as the preferred means of fluid resuscitation in trauma patients;, however, most combat units do not receive blood products prior to executing combat operations. This is largely due to logistical limitations in both blood supply and transfusion equipment. Further, the vast majority of medics are not trained in transfusion protocol. For many medics, the logistical constraints for cold-stored blood products favor the use of Walking Blood Bank (WBB), however few cases have been reported of WBB implementation at the point of injury during real world combat operations. This case report reviews one case of successful transfusion using WBB procedures at point of injury during combat. It highlights not only the feasibility, but also the necessity, for implementation of this practice on a larger scale.
| Tags : transfusion
20/01/2022
Fluid Resuscitation in Tactical Combat Casualty Care. Update 201
Fluid Resuscitation in Tactical Combat Casualty Care
TCCC Guidelines Change 21-01
4 November 2021
Dans ce document le mot important est choc hémorragique. Ce n'est pas parce q'un blessé saigne qu'il est en état de choc.
| Tags : choc, hémorragie, transfusion
17/07/2021
Transfuser avant l'hôpital: Pas suffisant pour réduire la mortalité
Effect of Prehospital Red Blood Cell Transfusion on Mortality and Time of Death in Civilian Trauma Patients
Background:
Current management principles of hemorrhagic shock after trauma emphasize earlier transfusion therapy to prevent dilution of clotting factors and correct coagulopathy. London's Air Ambulance (LAA) was the first UK civilian prehospital service to routinely offer prehospital red blood cell (RBC) transfusion (phRTx). We investigated the effect of phRTx on mortality.
Methods:
Retrospective trauma database study comparing mortality before implementation with after implementation of phRTx in exsanguinating trauma patients. Univariate logistic regression was performed for the unadjusted association between phRTx and mortality was performed, and multiple logistic regression adjusting for potential confounders.
Results:
We identified 623 subjects with suspected major hemorrhage. We excluded 84 (13.5%) patients due to missing data on survival status. Overall 187 (62.3%) patients died in the before phRTx period and 143 (59.8%) died in the after phRTx group. There was no significant improvement in overall survival after the introduction of phRTx (P = 0.554). Examination of prehospital mortality demonstrated 126 deaths in the pre-phRTx group (42.2%) and 66 deaths in the RBC administered group (27.6%). There was a significant reduction in prehospital mortality in the group who received RBC (P < 0.001).
Conclusions:
phRTx was associated with increased survival to hospital, but not overall survival. The “delay death” effect of phRTx carries an impetus to further develop inhospital strategies to improve survival in severely bleeding patients.
23/02/2021
Hypocalcémie ? Possible avant toute transfusion
Hypocalcemia in Military Casualties From Point of Injury to Surgical Teams in Afghanistan
Conner JR et Al. Mil Med . 2021 Jan 25;186(Suppl 1):300-304. doi: 10.1093/milmed/usaa267
---------------------------------------------------
On sait que les dérivés sanguins conservés en solution citratée exposent à une hypocalcémie lors de transfusions importantes, surtout en cas de traumatismes secondaires à des explosions. Apparemment cette dernière peut aussi survenir avant la mise en oeuvre de transfusion. C'est ce que laisse à penser ce document qui interpelle quand au rôle de la survenue d'une hypocalcémie (Ca2+ionisé) en phase préhospitalière. Quid de la validité des travaux ayant porté sur la transfusion hospitalière sans contrôle de ce paramètre Calcium ? Au TXA, faut il ajouter le Ca2+ ?
---------------------------------------------------
Hypocalcemia is a known sequela of citrated blood product transfusion. Civilian data suggest hypocalcemia on hospital admission is associated with worse outcomes. Initial calcium levels in military casualties have not previously been analyzed. The objective of this retrospective review aimed to assess the initial calcium levels in military trauma casualties at different Forward Surgical Teams (FST) locations in Afghanistan and describe the effects of prehospital blood product administration on arrival calcium levels.
This is a retrospective cohort analysis of military casualties arriving from point of injury to one of two FSTs in Afghanistan from August 2018 to February 2019 split into four locations. The primary outcome was incidence of hypocalcemia (ionized calcium < 1.20 mmol/L).
There were 101 patients included; 55 (54.5%) experienced hypocalcemia on arrival to the FST with a mean calcium of 1.16 mmol/L (95% confidence interval [CI], 1.14 to 1.18). The predominant mechanism of injury consisted of blast patterns, 46 (45.5%), which conferred an increased risk of hypocalcemia compared to all other patterns of injury (odds ratio = 2.42, P = .042).

Thirty-eight (37.6%) patients required blood product transfusion. Thirty-three (86.8%) of the patients requiring blood product transfusion were hypocalcemic on arrival. Mean initial calcium of patients receiving blood product was 1.13 mmol/L (95% CI, 1.08 to 1.18), which was significantly lower than those who did not require transfusion (P = .01). Eight (7.9%) of the patients received blood products before arrival, with 6/8 (75%) presenting with hypocalcemia.
Hypocalcemia develops rapidly in military casualties and is prevalent on admission even before transfusion of citrated blood products. Blast injuries may confer an increased risk of developing hypocalcemia. This data support earlier use of calcium supplementation during resuscitation.
15/11/2020
Solutés hypersalés: Sûrs pour la coagulation
The effect of hypertonic saline and mannitol on coagulation in moderate traumatic brain injury patients
Wang H et Al. Am J Emerg Med. 2017 Oct;35(10):1404-1407
-------------------------------------
Du moins pour le salé à 3%, administré ici pendant 3 jours.
-------------------------------------
Background:
Hyperosmolar therapy, using either hypertonic saline (HTS) or mannitol (MT), is considered the treatment of choice for intracranial hypertension, a disorder characterized by high intracranial pressure (ICP). However, hyperosmolar agents have been postulated to impair coagulation and platelet function. The aim of this study was to identify whether HTS and MT could affect coagulation in moderate traumatic brain injury (TBI) patients.
Methods: In this prospective and randomized double-blind study, we included adult patients with moderate TBI. Patients were divided into two groups according to the type of hypertonic solution administered. Group A patients received 20% MT and group B patients received 3% HTS. Rotational thromboelastometry (ROTEM) parameters were used to assess coagulation and platelet function.
Results:
ROTEM parameters included CT (clotting time), CFT (clot formation time), maximum clot firmness (MCF) measured by MCF (EXTEM and INTEM), MCF (FIBTEM) and standard coagulation tests (p>0.05). No significant differences were found between the two groups. Moreover, ROTEM parameters did not show significant changes at different time points after administration of the hyperosmolar solutions (p>0.05).
Conclusions: Overall, use of 3% HTS and 20% MT for the control of ICP did not significantly affect patients' coagulation function. Therefore, hyperosmotic solution is safe and does not increase the risk of intracranial rebleeding.