12/11/2015
Lent/régulier: Mieux pour le crâne ?
Resuscitation speed affects brain injury in a large animal model of traumatic brain injury and shock
Sillesen M et Al. Scand J Trauma Resusc Emerg Med. 2014 Aug 14;22:46
----------------------------------------
En matière de remplissage vasculaire, on s'intéresse souvent à la nature des solutés et aux quantités perfusées. On s'intéresse moins aux vitesses de perfusion. Pourtant cette dernière a son importance. Le travail présenté est intéressant car il rapport qu'outre la nature du soluté importante mais aussi sa vitesse d'administration. Ainsi il semblerait que dans un modèle expérimental de traumatisme crânien l'emploi de solutés cristalloïdes de sérum salé isotonique soit responsable d'une augmentation de l'oedeme cérébal et du volume des lésions intracraniennes quel que soit le régime d'administration. Ce travail retrouve par ailleurs l'intérêt sur la maîtrise de l'oedeème cérébral de l'apport de fraction coagulantes de manière procédurée par rapport au bolus. Si l'on replace ce document dans le contexte d'isolement et de maintien en survie de nos blessés notamment cranio-cérébraux en état de choc, cela implique une formation spécifique des équipes qui devront par ailleurs pouvoir disposer d'équipements adaptés comme celui_ci (1)
----------------------------------------
BACKGROUND:
Optimal fluid resuscitation strategy following combined traumatic brain injury (TBI) and hemorrhagic shock (HS) remain controversial and the effect of resuscitation infusion speed on outcome is not well known. We have previously reported that bolus infusion of fresh frozen plasma (FFP) protects the brain compared with bolus infusion of 0.9% normal saline (NS). We now hypothesize reducing resuscitationinfusion speed through a stepwise infusion speed increment protocol using either FFP or NS would provide neuroprotection compared with a highspeed resuscitation protocol.
METHODS:
23 Yorkshire swine underwent a protocol of computer controlled TBI and 40% hemorrhage. Animals were left in shock (mean arterial pressure of 35 mmHg) for two hours prior to resuscitation with bolus FFP (n = 5, 50 ml/min) or stepwise infusion speed increment FFP (n = 6), bolus NS (n = 5, 165 ml/min) or stepwise infusion speed increment NS (n = 7). Hemodynamic variables over a 6-hour observation phase were recorded. Following euthanasia, brains were harvested and lesion size as well as brain swelling was measured.
RESULTS:
Bolus FFP resuscitation resulted in greater brain swelling (22.36 ± 1.03% vs. 15.58 ± 2.52%, p = 0.04), but similar lesion size compared with stepwise resuscitation. This was associated with a lower cardiac output (CO: 4.81 ± 1.50 l/min vs. 5.45 ± 1.14 l/min, p = 0.03). In the NS groups, bolus infusion resulted in both increased brain swelling (37.24 ± 1.63% vs. 26.74 ± 1.33%, p = 0.05) as well as lesion size (3285.44 ± 130.81 mm(3) vs. 2509.41 ± 297.44 mm3, p = 0.04). This was also associated with decreased cardiac output (NS: 4.37 ± 0.12 l/min vs. 6.35 ± 0.10 l/min, p < 0.01).
CONCLUSIONS:
In this clinically relevant model of combined TBI and HS, stepwise resuscitation protected the brain compared with bolusresuscitation.

| Tags : tbi, remplissage
10/11/2015
Dépakine chez le blessé cranien en choc ?
Treatment with a histone deacetylase inhibitor, valproic acid, is associated with increased platelet activation in alarge animal model of traumatic brain injury and hemorrhagic shock
Dekker SE et Al. J Surg Res. 2014 Jul;190(1):312-8
---------------------------
Le concept du damaged control resuscitation fait appel en partie à de nouvelles modalités transfusionnelles et d'emploi de fractions coagulantes. D'autres approches sont possibles comme celles visant à restuarer la fonction plaquettaire. C'est ce que permettrait l'adminsitration de médicaments appartenant à la classe des inhibiteurs des histone deacetylase et dont les effets neuroprotecteurs pourraient ainsi être mis à profit. Le document proposé semble conforter cette approche.
---------------------------
BACKGROUND:
We have previously shown that resuscitation with fresh frozen plasma (FFP) in a large animal model of traumatic brain injury (TBI) and hemorrhagic shock (HS) decreases the size of the brain lesion, and that addition of a histone deacetylase inhibitor, valproic acid (VPA), provides synergistic benefits. In this study, we hypothesized that VPA administration would be associated with a conservation of platelet function as measured by increased platelet activation after resuscitation.
MATERIALS AND METHODS:
Ten swine (42-50 kg) were subjected to TBI and HS (40% blood loss). Animals were left in shock for 2 h before resuscitation with either FFP or FFP+VPA (300 mg/kg). Serum levels of platelet activation markers transforming growth factor beta, CD40 L, P-selectin, and platelet endothelial cell adhesion molecule (PECAM) 1 were measured at baseline, postresuscitation, and after a 6-h observation period. Platelet activation markers were also measured in the brain whole cell lysates and immunohistochemistry.
RESULTS:
Circulating P-selectin levels were significantly higher in the FFP+VPA group compared with the FFP alone group (70.85±4.70 versus 48.44±7.28 ng/mL; P<0.01). Likewise, immunohistochemistry data showed elevated P-selectin in the VPA treatment group (22.30±10.39% versus 8.125±3.94%, P<0.01). Serum sCD40L levels were also higher in the FFP+VPA group (3.21±0.124 versus 2.38±0.124 ng/mL; P<0.01), as was brainsCD40L levels (1.41±0.15 versus 1.22±0.12 ng/mL; P=0.05). Circulating transforming growth factor beta levels were elevated in the FFP+VPA group, but this did not reach statistical significance (11.20±1.46 versus 8.09±1.41 ng/mL; P=0.17). Brain platelet endothelial cell adhesion molecule 1 levels were significantly lower in the FFP+VPA group compared with the FFP group (5.22±2.00 pg/mL versus 7.99±1.13 pg/mL; P=0.03).
CONCLUSIONS:
In this clinically relevant large animal model of combined TBI+HS, the addition of VPA to FFP resuscitation results in an early upregulation of platelet activation in the circulation and the brain. The previously observed neuroprotective effects of VPA may be due to a conservation of platelet function as measured by a higher platelet activation response after resuscitation.
| Tags : tbi, coagulopathie
11/01/2015
Histoire de casque de protection
Ballistic helmets – Their design, materials, and performance against traumatic brain injury
Kulkarni SG et Al. Composite Structures 101 (2013) 313–331
Protecting a soldier’s head from injury is critical to function and survivability. Traditionally, combat helmets have been utilized to provide protection against shrapnel and ballistic threats, which have reduced head injuries and fatalities. However, home-made bombs or improvised explosive devices (IEDs) have been increasingly used in theatre of operations since the Iraq and Afghanistan conflicts. Traumatic brain injury (TBI), particularly blast-induced TBI, which is typically not accompanied by external body injuries, is becoming prevalent among injured soldiers. The responses of personal protective equipment, especially combat helmets, to blast events are relatively unknown. There is an urgent need to develop head protection systems with blast protection/mitigation capabilities in addition to ballistic protection.
Modern military operations, ammunitions, and technology driven war tactics require a lightweight headgear that integrates protection mechanisms (against ballistics, blasts, heat, and noise), sensors, night vision devices, and laser range finders into a single system.
The current article provides a comparative study on the design, materials, and ballistic and blast performance of the combat helmets used by the US Army based on a comprehensive and critical review of existing studies. Mechanisms of ballistic energy absorption, effects of helmet curvatures on ballistic performance, and performance measures of helmets are discussed. Properties of current helmet materials (including Kevlar K29, K129 fibers and thermoset resins) and future candidate materials for helmets (such as nano-composites and thermoplastic polymers) are elaborated. Also, available experimental and computational studies on blast-induced TBI are examined, and constitutive models developed for brain tissues are reviewed. Finally, the effectiveness of current combat helmets against TBI is analyzed along with possible avenues for future research.
| Tags : balistique, blast, tbi
21/11/2013
Le plasma lyophilisé: Bon pour le cerveau du traumatisé qui saigne
Early treatment with lyophilized plasma protects the brain in a large animal model of combined traumatic brain injury and hemorrhagic shock
Imam AM et Al. J Trauma Acute Care Surg. 2013;75: 976-983
accéder aux abstracts de la WTA publiés dans J trauma Acute care
_____________________________________
Bien sûr une étude animale, mais une de plus qui milite pour un emploi précoce du plasma lyophylisé.
_____________________________________
BACKGROUND: Combination of traumatic brain injury (TBI) and hemorrhagic shock (HS) can result in significant morbidity and mortality. We have previously shown that early administration of fresh frozen plasma (FFP) in a large animal model of TBI and HS reduces the size of the brain lesion as well as the associated edema. However, FFP is a perishable product that is not well suited for use in the austere prehospital settings. In this study, we tested whether a shelf-stable, low-volume, lyophilized plasma (LSP) product was as effective as FFP.
METHODS:
Yorkshire swine (42-50 kg) were instrumented to measure hemodynamic parameters, intracranial pressure, and brain tissue oxygenation. A prototype, computerized, cortical impact device was used to create TBI through a 20-mm craniotomy: 15-mm cylindrical tipimpactor at 4 m/s velocity, 100-millisecond dwell time, and 12-mm penetration depth. Volume-controlled hemorrhage was induced(40-45% total blood volume) concurrent with the TBI. After 2 hours of shock, animals were treated with (1) normal saline (NS, n = 5), (2) FFP (n = 5), and (3) LSP (n = 5). The volume of FFP and LSP matched the shed blood volume, whereas NS was 3 times the volume. Six hours after resuscitation, brains were sectioned and stained with TTC (2, 3, 5-Triphenyltetrazolium chloride), and lesion size (mm3) and swelling (percent change in volume compared with the contralateral, uninjured side) were measured.
RESULTS:
This protocol resulted in a highly reproducible brain injury, with clinically relevant changes in blood pressure, cardiac output, tissue hypoperfusion, intracranial pressure, and brain tissue oxygenation. Compared with NS, treatment with LSP significantly ( p G 0.05) decreased brain lesion size and swelling (51% and 54%, respectively).
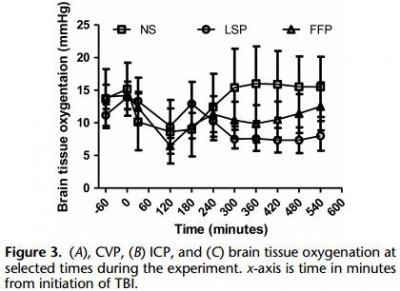
CONCLUSION: In a clinically realistic combined TBI + HS model, early administration of plasma products decreases brain lesion size and edema. LSP is as effective as FFP, while offering many logistic advantages.
| Tags : tbi, coagulopathie
13/09/2013
Psychological Health and Traumatic Brain Injury
25/06/2013
Blast TBI: Un seuil plus bas que pour le poumon
Working toward exposure thresholds for blast-induced traumatic brain injury: thoracic and acceleration mechanisms.
Courtney MW et all. Neuroimage, 2011 Jan,54 Suppl 1: S55-61
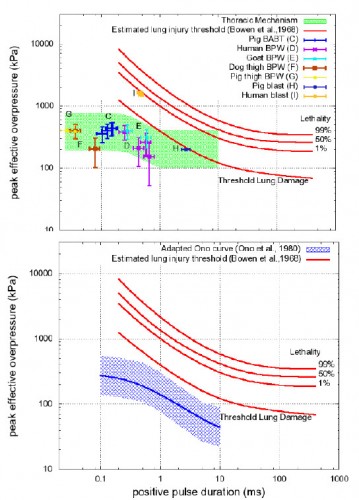
Les mécanismes du blast cérébral associent la tansmission de la surpression via le thorax, l'impact direct sur la boite crânienne et les phénomènes d'accélaration de l'extrémité céphalique. Ce document explore la première voie et exprime bien que les seuils lésionnels de lésions cérébrales (courbes de Ono) sont plus bas que les seuils pulmonaires(courbes de Bowden).
| Tags : balistique, tbi
13/09/2012
Trauma crânien et psychologique: Le point US
| Tags : ptsd, tbi, psychiatrie

