21/03/2026
Pansement Israélein: Interrêt de la barre de compression locale
17/03/2026
Délais de prise en charge allongés et pansement hémostatique: Danger

26/02/2026
Stabilisation pelvienne par garrot tourniquet: Parait OK
Pst Hémostatique et temérpatures extrêmes: C'est OK
13/02/2026
Hémorragie des blessés de guerre: Quoi de disponible ?
--------------------------------
12/02/2026
Hydrogel hémostatique: Cela arrive
The next frontier in hemorrhagic management: A comprehensive review on development of natural polymer-based injectable hydrogels as promising hemostatic dressings
Sanya A et Al. Chemical Engineering Journal 10.1016/j.cej.2024.155033
-----------------------------
L'idée d'injecter un produit hémostatique (poudre, petites épnges, gel) au contact d'une lésion qui saigne n'est pas nouvelle et de nombreux travaux de recherche sont en train de paraître dans la littérature (1, 2). Cet article fait une courte synthèse des enjeux de ce type de produit qui paraissent bien complémentaire des moyens mécanique d'arrêt des hémorragies.
-----------------------------
Unmet needs in controlling uncontrolled hemorrhage in emergency, surgical, and battlefield scenarios have sparked widespread interest in developing new hemostatic technologies. Despite the availability of a wide range of commercially available hemostats, sealants, and adhesives, the development of ideal hemostatic formulations with remarkable properties such as the ability to promptly staunch bleeding under wet and dynamic conditions remains a challenge. Injectable polymeric hydrogels with superior hemostatic qualities besides their irregular wound filling capability, and minimal invasiveness have received considerable interest in recent years because of their attractive properties particularly useful in bleeding management. In this article, the most recent advances in the fabrication of injectable hydrogel systems from natural polymers towards hemostatic applications are discussed based on an extensive review of the literature. Additionally, the inherent hemostatic attributes of each polymer have been highlighted alongwith a comprehensive overview of various mechanisms through which injectable hydrogels achieve hemostasis. Furthermore, the challenges that need to be tackled towards accelerating the translation of these novel hemostatic hydrogel systems to clinical practice are emphasized and future directions for research in the field are also presented.
Parmi ces produits: le TraumaGel
11/02/2026
Garrot mieux que les autres moyens? Et bien pas si sûr
Is the Use of Tourniquets More Advantageous than Other Bleeding Control Techniques in Patients with Limb Hemorrhage? A Systematic Review and Meta-Analysis
Cirocchi R et Al. Medicina (Kaunas). 2025 Jan 9;61(1):93
--------------------------
Une méta analyse qui jette un peu d'ombre sur le sacro-saint tourniquet, surtout quand il n'est pas bien utilisé. Un retour vers les pansements compressifs ? Probablement pas? surtout surveiller ces garrots et savoir les convertir correctement.
--------------------------
Background and Objectives:
Trauma, particularly uncontrolled bleeding, is a major cause of death. Recent evidence-based guidelines recommend the use of a tourniquet when life-threating limb bleeding cannot be controlled with direct pressure. Prehospital hemorrhage management, according to the XABCDE protocol, emphasizes the critical role of tourniquets in controlling massive bleeding. The aim of this systematic review and meta-analysis was to summarize data from the available scientific literature on the effectiveness of prehospital tourniquet use for extremity bleeding.
Materials and Methods:
A systematic review and meta-analysis was performed between March 2022 and March 2024, adhering to PRISMA guidelines, to determine whether prehospital tourniquets are clinically effective. The protocol was published on PROSPERO (ID number: CRD42023450373).
Results:
A comprehensive literature search yielded 925 articles and 11 studies meeting the inclusion criteria. The analysis showed a non-statistically significant reduction in mortality risk with tourniquet application (4.02% vs. 6.43%, RR 0.70, 95% CI 0.46-1.07). Analysis of outcomes of amputation of the traumatized limb indicated a statistically higher incidence of initial amputation in the tourniquet group (19.32% vs. 6.4%, RR 2.07, 95% CI 1.21-3.52), while delayed amputation showed no difference (9.39% vs. 3.66%, RR 0.93, 95% CI 0.42-2.07). Tourniquet use demonstrated a non-significant reduction in the number of blood components transfused (MD = -0.65; 95% CI -5.23 to 3.93 for pRBC, MD = -0.55; 95% CI -4.06 to 2.97 for plasma).
Conclusions:
Despite increasing use in civilian settings, this systematic review and meta-analysis showed no significant reduction in mortality or blood product use associated with prehospital tourniquet use. Further research, including high-quality randomized controlled trials, is required, as well as awareness and education campaigns relating to proper tourniquet use in the prehospital setting.
| Tags : tourniquet
16/12/2025
Quoi d'autre que le sang pour le choc hémorragique: ?
Adjuvant therapies for management of hemorrhagic shock: a narrative review
Daniel Y et Al. Crit Care. 2025 Mar 29;29(1):138
BackgroundSevere bleeding remains a leading cause of death in patients with major trauma, despite improvements in care during the acute phase, especially the application of damage control concepts. Death from hemorrhage occurs rapidly after the initial trauma, in most cases before the patient has had a chance to reach a hospital. Thus, the development of adjuvant drugs that would increase the survival of injured patients is necessary. Among the many avenues of research in this area, one is to improve cell survival during tissue hypoxia. During hemorrhagic shock, oxygen delivery to cells decreases and, despite increased oxygen extraction, anaerobic metabolism occurs, leading to acidosis, coagulopathy, apoptosis, and organ dysfunction.
Methods
We selected six treatments that may help cells cope with this situation and could be used as adjuvant therapies during the initial resuscitation of severe trauma patients, including out-of-hospital settings: niacin, thiazolidinediones, prolyl hydroxylase domain inhibitors, O-GlcNAcylation stimulation, histone deacetylase inhibitors, and adenosine–lidocaine–magnesium solution. For each treatment, the biological mechanism involved and a systematic review of its interest in hemorrhagic shock (preclinical data and human clinical trials) are presented.

Conclusion
Promising molecules, some of which are already used in humans for other indications, give us hope for human clinical trials in the field of hemorrhagic shock in the near future
07/12/2025
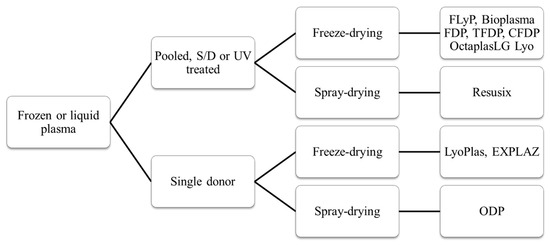
Vers un nouveau Plyo
Efficacy and safety of novel freeze-dried plasma products in a porcine combat casualty model
Dufour-Gaume F et Al. Transfusion. 2024 Sep;64(9):1670-1682
-----------
Le Plyo est un des produits phares développé par le CTSA. Les recherches continuent pour améliorer les caractérisques. C'est le sens de ce tavail qui porte sur du Plyo plus concentré ou enrichi en lyophylisat de plaquettes. Ce dernier produit parait particulièrement intéressant. l'industrie pharmaceutique n'est pas en reste puisqu'elle propose maintenant des alternatives au PLyo standard comme l'OctaplastLG considéré comme un médicament et non plus un dérivé du sang.
-----------
Background
Hemorrhagic shock is well documented as a leading cause of preventable fatalities among military casualties. During military operations plasma can be transfused while waiting for whole blood. This study was conducted to assess the safety and efficacy of two new freeze-dried plasma formulations in a porcine model of traumatic hemorrhagic shock.
Study Design and Methods
In the face of species-specific transfusion, transfusible blood products were derived from porcine sources. The efficacy of three lyophilized plasma (LP) formulations was evaluated: lyophilized plasma (LP), concentrated lyophilized plasma (CLP), and platelet-rich concentrated lyophilized plasma (PCLP). Pigs were subjected to multi-trauma and hemorrhagic shock. Ninety minutes post-shock induction, the animals were treated with one of the three lyophilized products. Monitoring included systolic blood pressure and cardiac output. Point-of-care and laboratory diagnostic tests were used to assess renal function, real-time hemostasis (ROTEM), and coagulation. Histological examinations of kidney, lung, and muscle tissues were conducted 4 h after shock induction.
Results
CLP and PCLP significantly improved systolic blood pressure and cardiac output and positively influenced base excess, creatinine, various ROTEM, and coagulation markers compared with standard LP without histologic modification. No adverse effect was associated with the transfusion of any of the plasma products throughout the experimental procedures.
Conclusion
Both CLP and PCLP exhibit promising therapeutic potential for managing hemorrhagic shock in scenario where whole blood supplies are limited. However, the distinct physiological and coagulation characteristics of the swine model necessitate further investigation using humanized preclinical models to fully understand their clinical applicability and constraints
Plasma: Un point en 2024
Dried Plasma for Major Trauma: Past, Present, and Future
05/12/2025
Point US sur l'emploi du TXA
Tranexamic acid in trauma: A joint position statement and resource document of NAEMSP, ACEP, and ACS-COT
Prehospital use of tranexamic acid (TXA) has grown substantially over the past decade despite contradictory evidence supporting its widespread use. Since the previous guidance document on the prehospital use of TXA for injured patients was published by the National Association of EMS Physicians, the American College of Surgeons Committee on Trauma, and the American College of Emergency Physicians in 2016, new research has investigated outcomes of patients who receive TXA in the prehospital setting. To provide updated evidence-based guidance on the use of intravenous TXA for injured patients in the emergency medical services (EMS) setting, we performed a structured literature review and developed the following recommendations supported by the evidence summarized in the accompanying resource document.
The National Association of EMS Physicians, the American College of Surgeons Committee on Trauma, and the American College of Emergency Physicians recommends:
• Prehospital TXA administration may reduce mortality in adult trauma patients with hemorrhagic shock when administered after lifesaving interventions.
• Prehospital TXA administration appears safe, with low risk of thromboembolic events or seizure.
• The ideal dose, rate, and route of prehospital administration of TXA for adult trauma patients with hemorrhagic shock has not been determined. Current evidence suggests EMS agencies may administer either a 1-g intravenous/intraosseous dose (followed by a hospital-based 1-g infusion over 8 hours) or a 2-g intravenous/intraosseous dose as an infusion or slow push.
• Prehospital TXA administration, if used for adult trauma patients, should be given to those with clinical signs of hemorrhagic shock and no later than 3 hours post-injury. There is no evidence to date to suggest improved clinical outcomes from TXA initiation beyond this time or in those without clinically significant bleeding.
• The role of prehospital TXA in pediatric trauma patients with clinical signs of hemorrhagic shock has not been studied, and standardized dosing has not been established. If used, it should be given within 3 hours of injury.
• Prehospital TXA administration, if used, should be clearly communicated to receiving health care professionals to promote appropriate monitoring and to avoid duplicate administration(s).
• A multidisciplinary team, led by EMS physicians, that includes EMS clinicians, emergency physicians, and trauma surgeons should be responsible for developing a quality improvement program to assess prehospital TXA administration for protocol compliance and identification of clinical complications.
30/11/2025
Garrot et grand froid: Possible ? ET bien OUI
Tourniquet self-application assessment in cold
weather conditions
Benítez YC et al. BMC Emergency Medicine (2023) 23:101
----------------------------
Le chox d'un garrot tactique n'est pas simple. Le CAT et le SOF-T sont parmi les plus répanduS. LE CoTCCC a évalué ses tourniquets et fait quelques propositions (1). Ces dernières ne prennet pas en compte certains besoins spécifiques comme leur emploi en zone grand froid ou en milieu maritime salin. C'est ce que propose de faire ce travail. Deux remarques: le SOFT-T ne fait pas partie des dispositifs testés. Le garrot OMNA semble être celui dont l'emploi est le plus facile dans ce contexte. Il ne s'agit pas d'un tourniquet mais d'un garrot à crématillère.
----------------------------
Background
Our study aimed to assess the ability of nonmedical civilians to self-apply extremity tourniquets in coldv weather conditions while wearing insulating technical clothing after receiving basic training.
Methods
A field study was conducted among 37 voluntary participants of an expedition party to the Spanish Antarctic base. The researchers assessed the participant’s ability to self-apply five commercial extremity tourniquets (CAT, OMNA, RMT, SWAT-T, and RATS) over cold-weather clothing and their achieved effectiveness for vascular occlusion. Upper extremity self-application was performed with a single-handed technique (OHT), and lower extremity applying a two-handed technique (THT). Perceptions of self-application ease mean values±standard
deviation (SD) were compared by applying a 5% statistical significance threshold. Frequency count determined tourniquet preference.
Results
All the tested ETs, except the SWAT-T, were properly self-applied with an OHT, resulting in effective vascular occlusion in the upper extremity. The five devices tested were self-applied correctly in the lower extremities using THT.
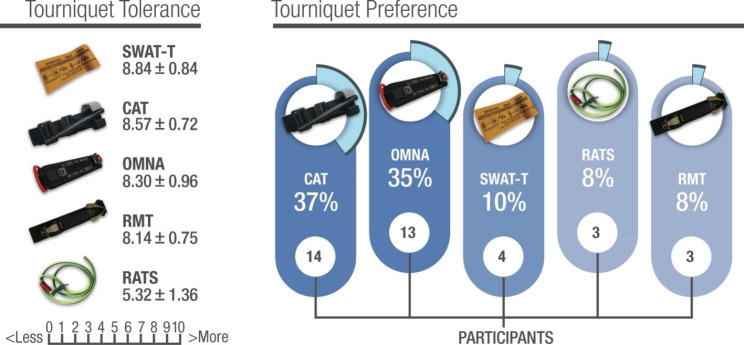
The ratcheting marine-designed OMNA ranked the highest for application easiness on both the upper and lower extremities, and the windlass CAT model was the preferred device by most participants.
Conclusions
Civilian extremity tourniquet self-application on both upper and lower extremities can be
accomplished in cold weather conditions despite using cold-weather gloves and technical clothing after receiving brief training. The ratcheting marine-designed OMNA ranked the highest for application ease, and the windlass CAT model was the preferred device
04/07/2025
Stabilisation pelvienne: Deux tourniquets font très bien
Assessment of proprietary and improvised pelvic binders: time to application and displacement during casualty evacuation
Howe TJ, et al. BMJ Mil Health 2025;0:1–5.
Introduction
Pelvic injuries resulting from high-energy trauma have an approximately 10% mortality rate. Suspected pelvic injuries are treated with pelvic binders to stabilise fractured bones and promote tamponade until surgical treatment. The effectiveness of pelvic binders in reducing mortality risk depends on accurate positioning of the binder. This study quantifies the ability of proprietary and improvised pelvic binders to maintain their positioning during a simulated casualty evacuation.
Methods
One improvised and three proprietary pelvic binders were tested in their ability to maintain their initial placement. Combat Medical Technicians applied binders to healthy subjects and then performed a simulated casualty evacuation. The time taken to apply each binder was measured. The evacuation consisted of: 20 m casualty drag, transfer onto a soft stretcher, 100 m evacuation on a soft stretcher, transfer onto a rigid stretcher and transfer into a field ambulance. Binder placement was measured using bony landmarks after the initial positioning and after each phase of the simulated casualty evacuation.
Results
The field-expedient pelvic splint (FEPS), SAM Pelvic Sling II (SAM) and T-POD Pelvic Stabilisation Device (TPOD) all remained within 45 mm of vertical displacement from their initial placement, which is considered an acceptable range for optimal binder functionality. The Prometheus Pelvic Splint (PROM) fell within this range in 83% of trials. The SAM was the fastest binder to apply, followed by the TPOD, and then the FEPS and PROM which took similar times to apply.
Conclusions
Binders were mostly able to maintain their positioning during the simulated casualty evacuations carried out in this study but differed in their application times. The improvised binder (FEPS) performed comparably to the proprietary binders tested, and its low cost and weight make it a good alternative to proprietary binders in the austere environment.
21/06/2025
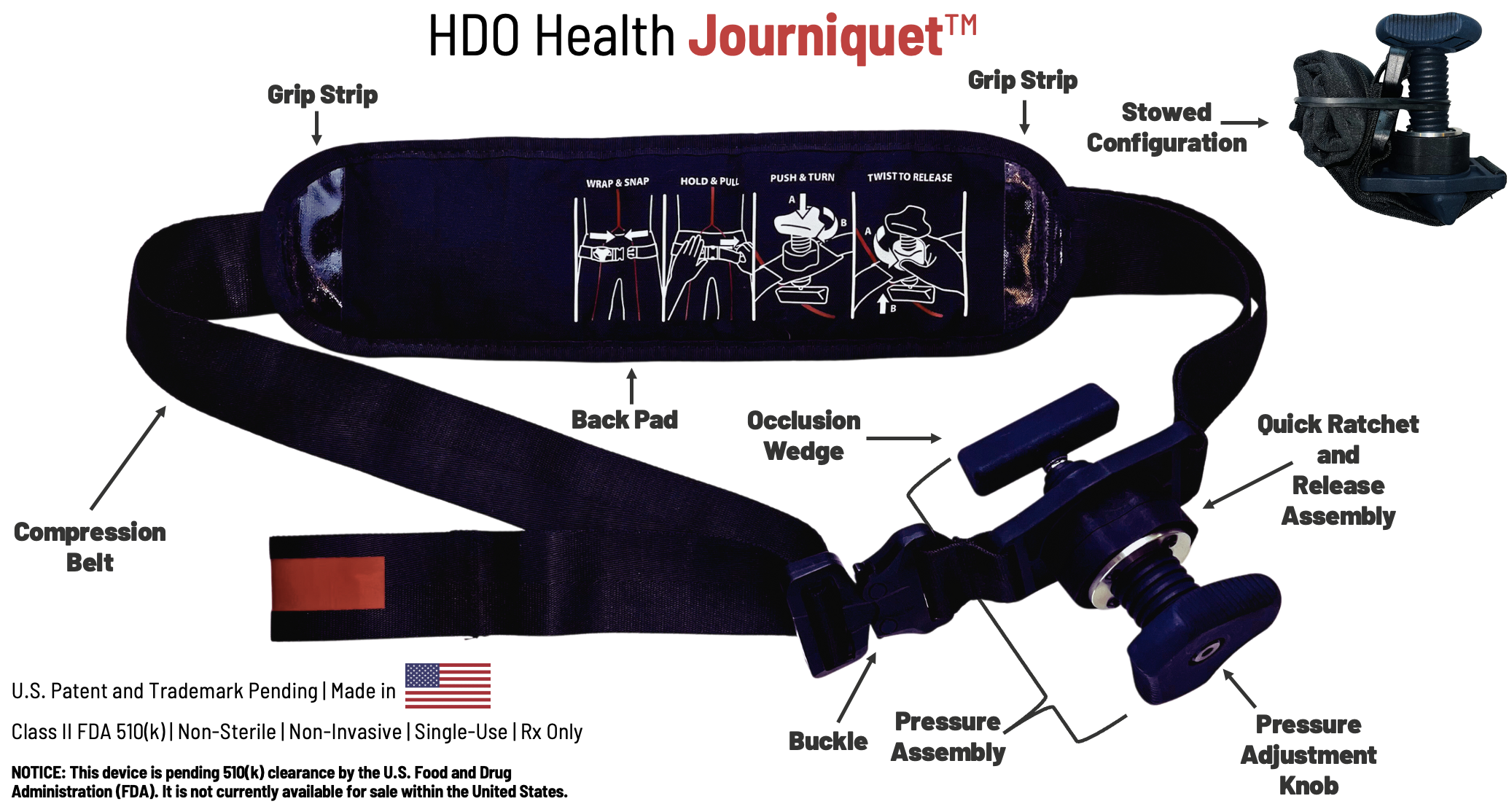
Tourniquet jonctionnel: Un nouveau venu, le Journiquet
01/05/2025
Tourniquet Abdominal: Probablement très utile.
Successful Management of Battlefield Traumatic Cardiac Arrest Using the Abdominal Aortic and Junctional Tourniquet (AAJT): A Case Series
Androshchuk D et Al. Case Reports J Spec Oper Med. 2025 Apr 4;25(1):65-69.
The Russo-Ukrainian war's prolonged warfare, resource constraints, and extended evacuation times have forced significant adaptations in Ukraine's medical system - including technological advancements and strategic resource placement. This study examined if the Abdominal Aortic and Junctional Tourniquet - Stabilized (AAJT-S) could manage traumatic cardiac arrest (TCA) at forward surgical stabilization sites (FSSS) as an adjunct to damage control surgery. Six patients in severe hypovolemic shock presented at an FSSS during fighting in Bakhmut (July 2022) and Slovyansk (May 2023). Following TCA due to exsanguination, the AAJT-S was applied 2cm below the umbilicus. Cardiopulmonary resuscitation (CPR) and transfusion (blood and/or plasma) were initiated. All six patients were resuscitated. None required vasopressor support post-resuscitation. Five survived to the next level of care. One died awaiting evacuation, and another of wounds after 10 days. Four survived to discharge. Three were followed and neurologically intact, and no death records matched the fourth's name and date of birth at 18 months. Follow-up was limited, but one patient was neurologically intact at one year. The AAJT-S effectively resuscitated TCA patients. It increased mean arterial pressure, focused resuscitative efforts on the upper torso, simplified care, and preserved crucial field resources. An alternative to traditional emergency thoracotomy, AAJT-S could replace or complement resuscitative endovascular balloon occlusion of the aorta in pre-hospital settings, given its ease of application by combat medics. AAJT-S, alongside blood transfusion and CPR, achieved 100% success in return of spontaneous circulation and effectively managed TCA in a wartime FSSS.
11/02/2025
Effets du garrot: temps dépendant !
Timeline effects of tourniquets used in trauma care
Pascoe B et Al. JHTAM. 2024;6(2):26-32
"After reviewing current evidence, having evacuation protocols which ensure tourniquets are only required to be applied for an upper window of two hours would be a safe guideline to work with. Prioritising the evacuation of patients to ensure they are moved to a facility where tourniquets are able to be removed within the upper limit of two hours, would minimise the risk of serious co-morbidities and maximise the chance for full recovery. In addition to this, improving education on the correct use and application of the tourniquet would also improve patient outcomes."

17/01/2025
Conflit ukrainien: Pour de la chirurgie encore plus proche des combats
09/10/2024
REBOA préhospitalier: Attention !
Vascular complications secondary to resuscitative endovascular balloon occlusion of the aorta placement at a Level 1 Trauma Center
Tullos A et Al. J Vasc Surg. 2024 Jul;80(1):64-69
--------------------------------------------------------
Une technique dont on comprend l'intérêt théorique. Mais la vraie vie semble un peu différente. La mise en place d'un REBOA nécessite la présence d'une équipe chirurgicale très proche et ce n'est pas le cas le plus souvent en condition de combat.. L'environnement nécessaire au succès de cette technique n'est pas, le plus souvent, présent.
-------------------------------------------------------
Objective:
Resuscitative endovascular balloon occlusion of the aorta (REBOA) is designed to manage
severe hemorrhagic shock. Popularized in medical care during military conflicts, the concept has emerged as a lifesaving technique that is utilized around the United States. Literature on risks of REBOA placement, especially vascular injuries, are not well-reported. Our goal was to assess the incidence of vascular injury from REBOA placement and the risk factors associated with injury and death among these patients at our institution.
Methods:
We performed a retrospective cohort study of all patients who underwent REBOA placement between September 2017 and June 2022 at our Level 1 Trauma Center. The primary outcome variable was the presence of an injury related to REBOA insertion or use. Secondary outcomes studied were limb loss, the need for dialysis, and mortality. Data were analyzed using descriptive statistics, χ2, and t-tests asappropriate for the variable type.
Results:
We identified 99 patients who underwent REBOA placement during the study period. The mean
age of patients was 43.1 ± 17.2 years, and 67.7% (67/99) were males. The majority of injuries were from blunt trauma (79.8%; 79/99). Twelve of the patients (12.1%; 12/99) had a vascular injury related to REBOA placement. All but one required intervention. The complications included local vessel injury (58.3%; 7/12), distal embolization (16.7%; 2/12), excessive bleeding requiring vascular consult (8.3%; 1/12), pseudoaneurysm requiring intervention (8.3%; 1/12), and one incident of inability to remove the REBOA device (8.3%; 1/12). The repairs were performed by vascular surgery (75%; 9/12), interventional radiology (16.7%; 2/12), and trauma surgery (8.3%; 1/12). There was no association of age, gender, race, and blunt vs penetrating injury to REBOA-related complications. Mortality in this patient population was high (40.4%), but there was no association with REBOA-related complications. Ipsilateral limb loss occurred in two patients with REBOA-related injuries, but both were due to their injuries and not to REBOA-related ischemia.
Conclusions:
Although vascular complications are not unusual in REBOA placement, there does not appear to be an association with limb loss, dialysis, or mortality if they are addressed promptly. Close
coordination between vascular surgeons and trauma surgeons is essential in patients undergoing REBOA placement
Tourniquet en milieu froid: Des spécificités
Analysis of tourniquet pressure over military winter clothing and a short review of combat casualty care in cold weather warfare
Cold weather warfare is of increasing importance. Haemorrhage is the most common preventable cause of death in military conflicts. We analysed the pressure of the Combat Application Tourniquet® Generation 7 (CAT), the SAM® Extremity Tourniquet (SAMXT) and the SOF® Tactical Tourniquet Wide Generation 4 (SOFTT) over different military cold weather clothing setups with aleg tourniquet trainer. We conducted a selective PubMed search and supplemented this with own experiences in cold weather medicine. The CAT and the SAMXT both reached the cut off value of 180 mmHg in almost all applications. The SOFTT was unable to reach the 180 mmHg limit in less than 50% of all applications in some clothing setups. We outline the influence of cold during military operations by presenting differences between military and civilian cold exposure. We propose a classification of winter warfare and identify caveats and alterations of Tactical CombatCasualty Care in cold weather warfare, with a special focus on control of bleeding. The application of tourniquets over military winter clothing is successful in principle, but effectiveness may vary for different tourniquet models. Soldiers are more affected and impaired by cold than civilians. Militaryc ommanders must be made aware of medical alterations in cold weather warfare.
01/10/2024
REBOA préhospitalier: Une mortalité inchangée à 30 j, désillusion ?